Disclaimer: Even though biologically plausible, this protocol is speculative, though not risky.
Globally, over 400 million people have experienced long-COVID, which is about 1 in 20. The prevalence of long COVID seems to be 3-7%, which is huge. It is estimated that long COVID may be responsible for a loss of 1% of the world’s gross domestic product.
2 weeks ago, I caught COVID-19 for the 4th time. This time I likely caught it from a patient who I knew to be positive but had to insert a peripheral venous catheter. Even though I was wearing a mask, he started to cough multiple times very close to my face.
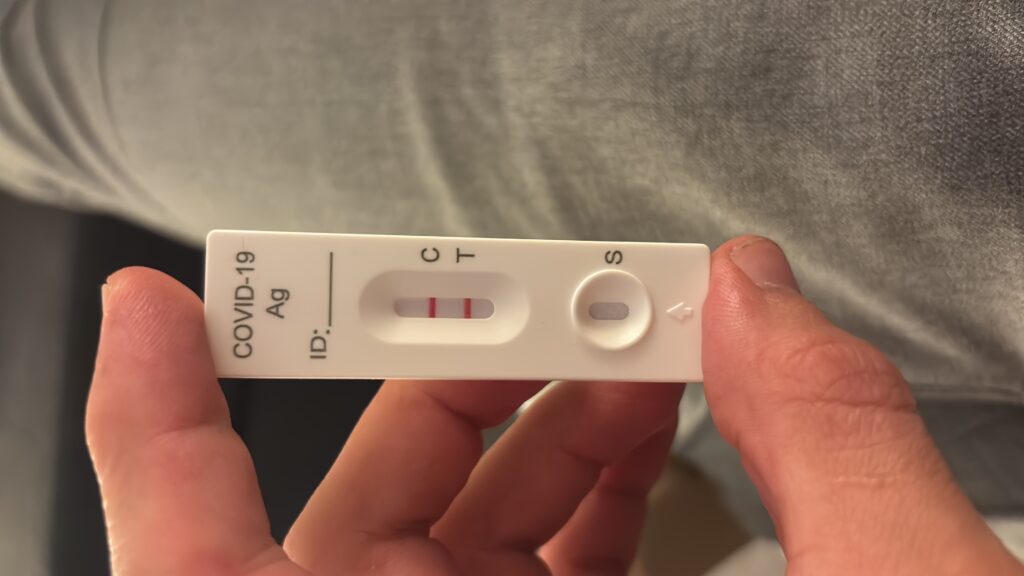
Like the two times before, I started my Long-COVID preventions protocol as soon as I tested positive. I published my old COVID prevention protocol here.
The disease period itself is usually relatively harmless for young healthy individuals. For example, fever, fatigue, etc. are all quite bearable compared to e.g., influenza, which keeps most people bedbound. However, Long-COVID is not.
I personally know a number of individuals whose lives have been absolutely derailed for many months to years by Long-COVID. One of my best friends is two years after and he is still having waxing and waning periods with symptoms. Another acquaintance had to quit his high-paying consulting job, his girlfriend left him, and he is now vegetating in his apartment. Another friend said that getting through Long-COVID without turning insane (his course lasted 3 years with many setbacks) is the biggest accomplishment in life. As I worked in the emergency apartment, I have also seen a couple of individuals whose life had been destroyed – unfortunately, as a doctor working in the conventional sick-care system I could not even try to help them (given that all my suggestions would have been “dangerous”, “experimental”, and off-label).

Anyway, the risk of contracting Long-COVID is about 2-4% overall per infection. Probably considerably lower after a mild non-first infection in a young healthy male (my guess is 1-2%). It is much higher in a non-mild infection and/or after the first infection.
Even though 1-2% does not sound like much, that is quite high, as this means that roughly 1 in 50 to 1 in a 100 people will get Long-COVID, even if they had been vaccinated, have had the virus already, and have a mild course. Estimates are that the UK has about 2 million individuals with lingering symptoms, which is a lot.
Specific early symptoms that recur in studies of people who later go on to develop long COVID: fatigue, headache, shortness of breath, loss of smell (anosmia), plus myalgia. However, long COVID has also been reported in asymptomatic individuals.
Long-COVID is pretty much the same as ME/CFS. ME/CFS, also known as chronic fatigue syndrome, includes a specific phenotype/illness that is sometimes found after infection with various viruses. The most documented of these is EBV. In fact, after mononucleosis, about 10% of infected people meet CFS criteria after 6 months, particularly if the infection happens in adulthood (vs. in childhood). Similarly, after the first COVID infection, about 10% (studies range from 5-20%) of people meet CFS criteria after 6 months. Other viruses for which this is reported are human herpes virus 6, CMV (another herpes virus), parvovirus B19, Ebola virus, and some enteroviruses.
What all of these viruses have in common is that they are slowly cleared by the body. Mononucleosis sometimes lasts for many weeks. Similarly, on the first COVID infection, people often test positive for weeks on end. The more severe and longer the infection the higher the likelihood of developing a CFS-like syndrome. A very simple formula that helps to predict the development of ME/CFS is disease severity times the time it takes until complete viral clearance.
This explains why CFS is very uncommon after influenza. While influenza is severe, the active illness duration is usually short and the body clears the virus relatively quickly, in contrast to, e.g., EBV and SARS-CoV-2.
After my first COVID infection four years ago, my CT value was positive/measurable for over 3 weeks. I was tired for about 1-2 months after, perhaps a very light version of LongCOVID, which I believe most people had (as most people cannot not tell whether they have 10-20% less energy for a couple of months).
In fact, even when flu symptoms feel intense (e.g., high fever), the median febrile phase is 3–5 days, viremia is minimal, and the immune system usually clears the virus quickly. By contrast, EBV has a prolonged, systemic, lymphoproliferative phase (weeks), and SARS-CoV-2 can also persist for weeks in tissues and is associated with delayed viral clearance in some. Duration of high cytokine activity seems to matter more than peak intensity. ME/CFS is strongly linked to aberrant immune reprogramming (chronic low-grade activation, poor NK cell function), which influenza rarely provokes. Furthermore, influenza mostly affects the respiratory epithelium whereas EBV and SARS-CoV-2 affect many other tissues (including neurons).
Individuals with CFS all have a similar phenotype. They have a pronounced fatigue, particularly after exertion (often lasting for days and weeks), also known as “crashes”, they often have a high heart rate, postural hypotension (dysautonomia), and a host of psychiatric symptoms (e.g., a tendency to be anxious). There also seems to be a host of other organ-specific symptoms.
A huge unanswered question is, why is ME/CFS more likely to develop if one has too much physical exertion during the recovery phase? For example, if people exercise too early, many long-COVID researchers agree that ME/CFS rates seem to be much higher. It seems that type-A personalities are overrepresented among CFS folks, presumably because they exercise too early when still sick. This could be explainable physiologically – e.g., exercise leading to increased IL-6 levels, exercise leading to injury to an already compromised endothelium, etc. But why the hell do people still report these exertion-induced crashes many months after the virus has already been cleared? For example, if people exercise intensely e.g., 6 months after contracting Long-COVID and before they have fully healed, many report that they get much worse again and stay worse for many weeks, also called post-exertional malaise (PEM). Is it because exercise temporarily increases cytokine levels and the increase in cytokine levels then signals to the brain that it should better stay in “shutdown” mode? I have no idea.
The protocol
The single biggest risk factor of developing Long-COVID is both the duration as well as the severity of the illness. Somebody who has a very short & mild course is much less likely to develop Long-COVID than somebody who has multiple symptoms for a longer amount of time. However, unfortunately, in absolute numbers, there are also a great many people who developed Long-COVID after a mild course. There are even some reports of people who developed long COVID after having been mostly asymptomatic.
Both the severity as well as the duration of the disease correlate well with viral load. This is where Paxlovid comes in. Paxlovid is a combination of two antivirals that slash viral load rapidly. According to the available data, paxlovid reduces the risk of Long-COVID by a bit – some studies find 20% others find less. However, there is no study that shows that Paxlovid increases long COVID risk, not even if people have a COVID rebound that happens in up to 20% of people who take Paxlovid.
As soon as I noticed symptoms, I started Paxlovid.
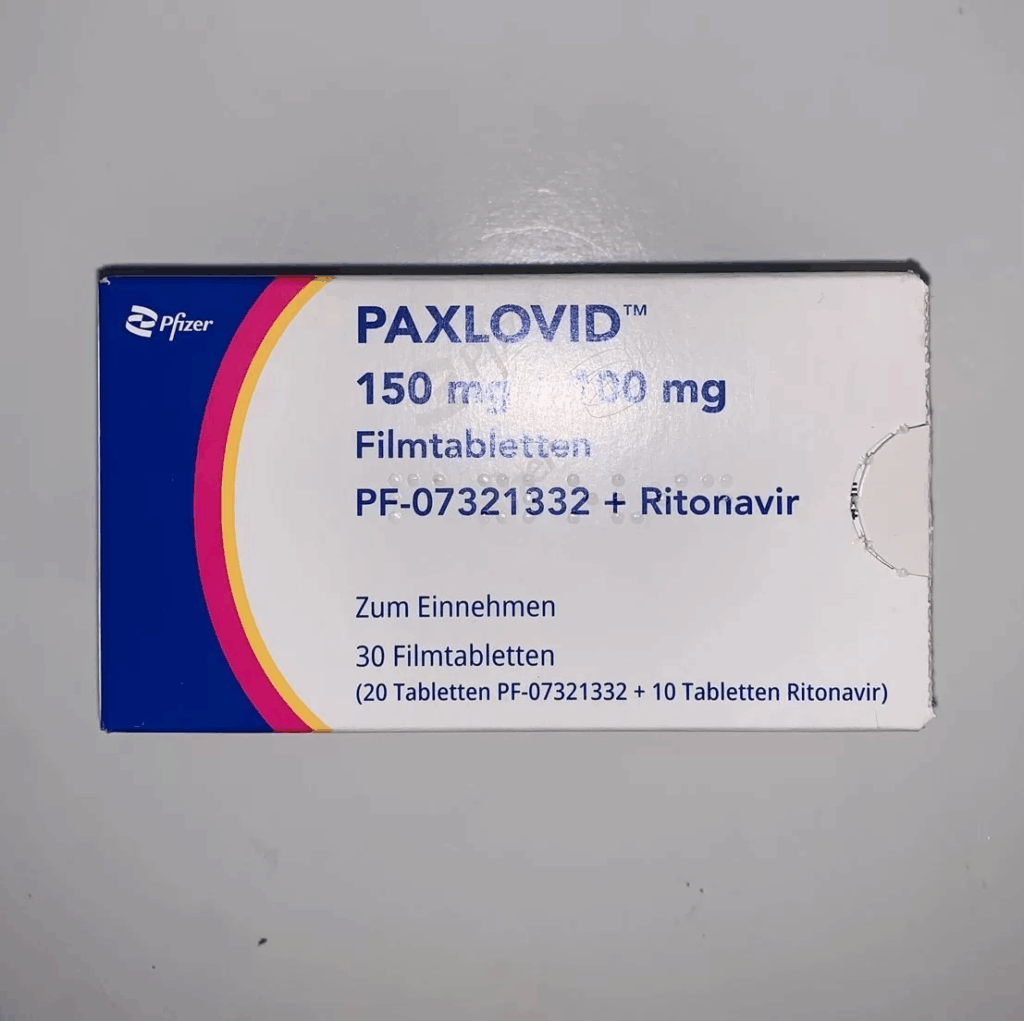
Like the times before, already 1 day after starting Paxlovid, I feel much better, probably representing a much reduced viral load. If my antigen test was strongly positive on day 1, it is only very faintly positive 24-36h after taking rapamycin.
Long-COVID is also associated with immune dysregulation and correlates with a high immune response (which sometimes is independent of viral load).
The last few times I had taken a single 3mg dose of rapamycin (which is boosted by the Paxlovid inhibiting CYP3A4). Rapamycin dampens my immune response. This could be a double-edged sword if I took it by itself because it may delay SARS-CoV-2 clearance a bit but it is great if combined with Paxlovid, which takes care of the viral replication. However, layering Paxlovid with rapamycin is quite risky because Paxlovid does increase sirolimus blood levels.
Matt Kaberlein (one of the leading longevity scientists) said in a podcast with Peter Attia that they have unpublished data (from online questionnaires of people taking rapamycin for longevity reasons) that long COVID rates are significantly lower in people who take rapamycin compared to longevity enthusiasts who do not. Surely there are confounders but at the very least the data does not go in the other direction.
There is the (theoretical?) risk that rapamycin taken early during a COVID infection may slow viral clearance.
Therefore, this time I had taken metformin. Metformin is the only drug that has RCT evidence that it lowers long COVID incidence, and not by a little. There are multiple trials that report that starting metformin within 3 days of symptom onset lowers long COVID risk by up to 40-60% (in overweight people). Mechanisms include mTOR inhibition, AMPK activation, and anti-inflammatory effects. How much the benefits are in a young, healthy, lean, insulin-sensitive male is unclear, but I would doubt that the benefits drop to zero.
In the trials they used 500mg of metformin on day one, 500mg twice per day on day 2-5, and 500mg three times per day on day 6 – day 14. Metformin was also associated with a faster viral clearance.
Metformin is ultra-low risk with a potentially large effect size to reduce long COVID risk. The biggest “risk” is gastrointestinal distress for 2 weeks or so. I take extended-release metformin, which has an even lower incidence of gastrointestinal issues.
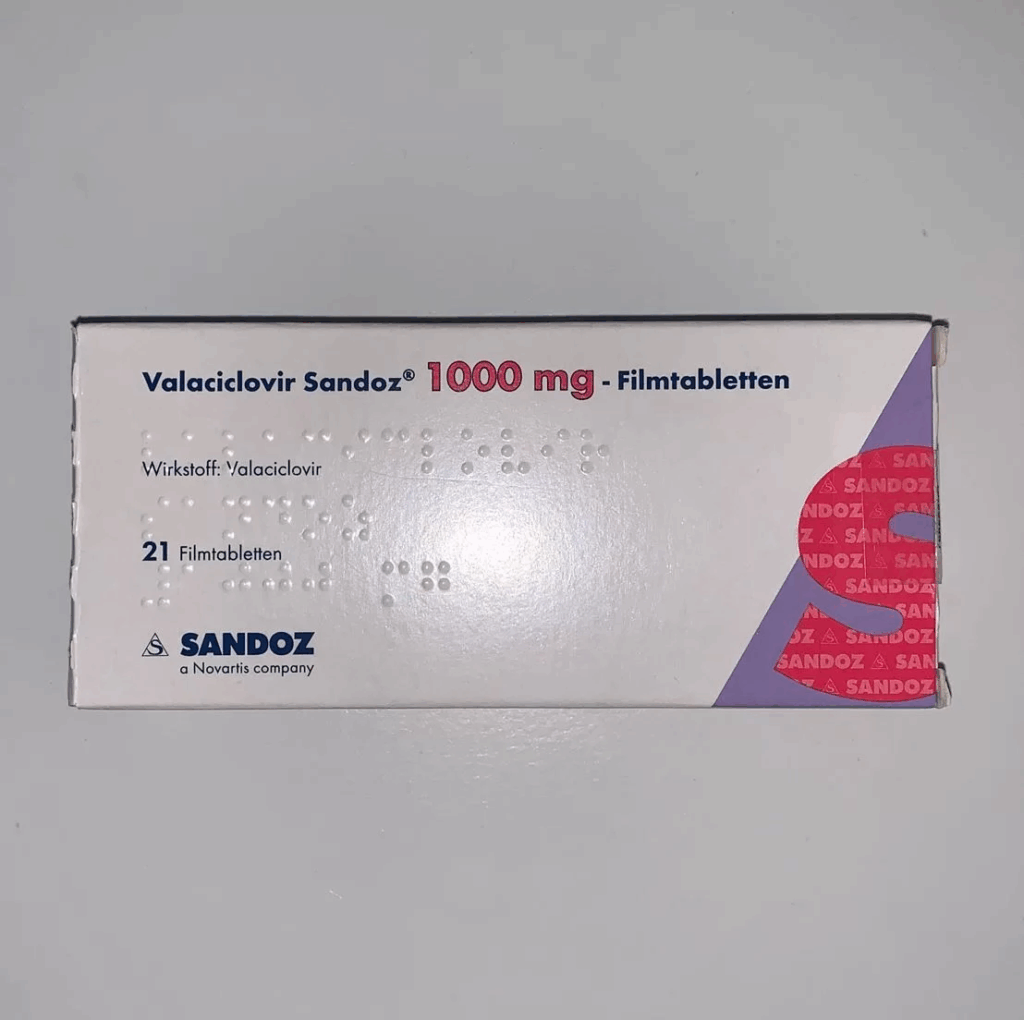
Long-COVID is also associated with reactivation of herpes viruses, particularly Epstein-Barr Virus (EBV), which is one of the most common causes of ME/CFS. Chronic fatigue syndrome has a strong overlap with Long-COVID . Because reactivation of EBV may drive up long COVID rates, I took 2x1g of valacyclovir on the first couple of days of the illness and then 500mg twice daily for 2 weeks. Similarly to metformin, this is pretty much negligible risk.
Some researchers argue that Long-COVID is also caused by the nervous system going into long-term “sickness” mode because of CNS inflammation, which then triggers a “shutdown”. This shutdown is partly induced by increased CNS cytokines and microglia activation and by reduced neuroplasticity – things that can be effectively counteracted with a single dose of ketamine.
In this vein, people with depression or mental distress (which is associated with neuroinflammation) have a 50% higher chance of going on to develop long COVID than non-depressed folks. Obviously, confounders abound but this association seems to hold.
The ketamine artificially boosts neuroplasticity (for about 7-14 days) during a vulnerable time period. There is also quite a bit of data on ketamine reducing neuroinflammation and microglia activation, and even on ketamine in Long-COVID. In my opinion, ketamine is best used for preventing Long-COVID (vs. treating it), if Long-COVID should indeed represent a CNS shutdown that is “imprinted” during the active phase of the disease. A low dose of ketamine is very low risk. At higher doses, ketamine is probably more neurotoxic than most people assume.
Anecdote time: A good friend of mine had COVID a couple of years ago. For 3 weeks after, he had severe brain fog and tiredness. At the time, he was completing his bachelor in physics and he had a big exam coming up. He said that for these 3 weeks after COVID, he sometimes was staring at his books and graphs for hours, but his brain was just foggy and his cognitive capabilities were mostly incapacitated. I pitched the ketamine and that it would be an experiment worth trying. High upside with low downside risk. I gave him 25mg subcutaneously. After about 24 hours (!) he claimed that his brain was back to normal and his brain fog had vanished, and it also did not return.
Ever since, I use 15-20mg of s.c. ketamine on day 5 after contracting COVID, when most of the symptoms have vanished. This reduces neuroinflammation and boosts neuroplasticity artificially for the next 2 weeks or so. Why such a low dose? Ritonavir is a strong inhibitor of CYP3A4 (and other CYP enzymes) which boosts the AUC of ketamine by unpredictably (1.5-3-fold). I weigh 80kg so in the worst case, the maximum dose I get is 45mg, which is close to the standard dose of 0.5mg/kg.
Summary
- I take Paxlovid to reduce viral load. Viral load is associated with disease severity. Disease severity is associated with Long-COVID. Data tells me that Paxlovid reduces my risk of contracting Long-COVID by a bit – how much is unlcear.
- I take metformin, which is the only drug that has RCT evidence to reduce long COVID development. Effect sizes seem to be quite large (40-60% in overweight individuals). Long-COVID is associated with immune dysregulation, particularly excessive cytokine levels. Metformin keeps an excessive immune response in check. Furthermore, metformin induces autophagy. Metformin is ultra low risk.
- I take valacyclovir because Long-COVID may be associated with the reactivation of endogenous herpes viruses. Valacyclovir has few to no risks.
- After symptoms are gone, I take a low dose of s.c. ketamine because Long-COVID is associated with CNS inflammation, microglial activation, and reduced neuroplasticity. Ketamine is well known to counteract all 3.
- I take REST because long COVID is associated with type A individuals who “push” during the active phase of their illness.
All this may seem overkill as the likelihood of contracting Long-COVID is less than 3% per infection. However, because some unlucky folks I know personally had their lives derailed by Long-COVID this is a potential 0-multiplier for months, which I am not willing to take.
Vaccination as a long COVID prevention
There is good data that getting a COVID vaccine rudes the risk of contracting COVID by about 50% for 3-6 months after vaccination. If one gets COVID despite vaccination, the illness is less severe and shorter, and the long COVID risk drops by about 30-50% (depending on the study). Thus, a vaccination reduces the risk of developing long COVID by roughly 2/3 for about 6 months or so after. So, from now on, I will get an additional vaccine shot every September, which should protect me quite a bit until springtime each year.
Sure, the vaccines are not optimal but they are a much smaller risk than the disease and its sequelae.
Bonus: Hydrocortisone
The days after COVID (or any other illness), I am a little more tired than usual. Part of this tiredness is due to HPA-axis downregulation, leading to lower cortisol levels. In fact, during a viral infection, cytokines drive up cortisol levels 2-5 fold. As the infection is cleared, for a couple of days up to a week, cortisol levels are lower than baseline, leading to (or at least contributing to) a “my batteries are half empty” feeling.
AUC-levels of cortisol are roughly 20-25mg per day in a healthy adult. A number of studies show that after an illness, cortisol levels are 20-30% lower, and this was also shown for COVID. By taking 5mg of hydrocortisone (roughly 25% of the daily need) in the morning for a couple of days after COVID, I feel and function much better. At this dose taken for a couple of days only, there is virtually no additional HPA-axis suppression.
Furthermore, there is data, that low cortisol levels are associated with long COVID development. Either this is a consquence of severe illness (severe illness has higher cortisol needs than mild illness) and thus just noise, or there is also causality because cortisol is dampening the immune system and low levels mean a hyperreactive immune system, which could be called immune dysregulation, which is associated with long COVID development.
I discuss my experience with hydrocortisone in more detail here.
Sources & further information
- Scientific article: Treatment of Long-Haul COVID Patients With Off-Label Acyclovir
- Scientific article: The potential of rapalogs to enhance resilience against SARS-CoV-2 infection and reduce the severity of COVID-19
- Scientific opinion article: Ketamine in COVID‐19 patients: Thinking out of the box
Disclaimer
The content available on this website is based on the author’s individual research, opinions, and personal experiences. It is intended solely for informational and entertainment purposes and does not constitute medical advice. The author does not endorse the use of supplements, pharmaceutical drugs, or hormones without the direct oversight of a qualified physician. People should never disregard professional medical advice or delay in seeking it because of something they have read on the internet.
