Multiple of my friends have tried a ketogenic diet. Most responded favorably, at least initially. Some had “through-the-roof” energy levels for the first couple of months, possibly in part related to a greatly increased cortisol secretion.
A couple of anecdotes. One friend even started a company shortly after starting the ketogenic diet, which he believed had given him the necessary energy levels. One friend, who has seasonal affective disorder, claimed that on the ketogenic diet, he did not have the winter blues he normally experiences. Two other friends claimed that the ketogenic diet helped their energy levels and mental clarity. Another friend claimed that going on keto helped his (self-diagnosed) autoimmune problems.
I personally responded great for a couple of months, but after that, I seemed to “burn out” as I did with intermittent fasting.
Unfortunately, when it comes to the promotion of different diets, one only hears about the cheerleaders making a lot of noise but not about the countless mute corpses in graves. What is more, many of the initial cheerleaders turn into corpses over time and then quiet down. So, informational supply about certain diets (e.g., keto, carnivore, intermittent fasting, vegan, etc.) is certainly biased.
The ketogenic diet alters a variety of physiological processes.
Whenever insulin levels are very low (such as during starvation, fasting, or very low-carbohydrate diets), lipolysis (the liberation of triglycerides from fat storage) is disinhibited and there is a strong flux of fatty acids out of adipocytes.
Low insulin also disinhibits hepatic ketogenesis, and a lot of the liberated fatty acids are converted into ketone bodies. Whenever ketone bodies rise above a certain threshold, this state is called “ketosis”.
Ketone bodies are basically “pre-digested” fatty acids with a chain length of four carbon atoms (vs. fatty acids usually have 14 to 20 carbon atoms). These small and water-soluble molecules can easily pass the blood-brain barrier and can be used for efficient energy generation by neurons, which under normal conditions primarily rely on glucose and lactate.
In addition to being a great fuel, ketone bodies are thought to have anti-inflammatory effects. Ketone bodies are also thought to directly or indirectly alter the expression of a number of genes.
Many people assume that most of the changes associated with a ketogenic diet come down to the rise in blood ketone levels. However, ketone bodies are only a fraction of the story.
The changes associated with a ketogenic diet are numerous, most of which are not a consequence of keeping blood ketone levels high but rather a consequence of keeping blood insulin levels very low. Insulin is the master control hormone of vertebrate metabolism. Among other things, insulin directly regulates the expression of about 2000 genes, which are presumably all altered at least to some extent on a ketogenic diet.
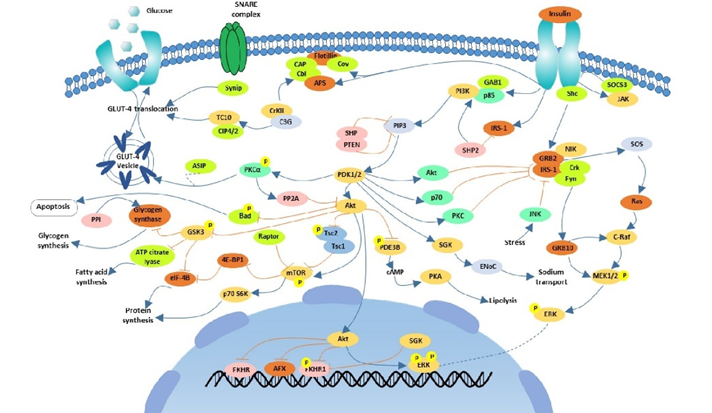
Insulin receptors are widely expressed throughout the brain, and there, insulin influences a wide variety of things, including endocrine regulation, monoamine levels (dopamine, noradrenaline, serotonin), sleep & appetite centers, and neurogenesis. Thus, some of the mental changes associated with a ketogenic diet are probably in part due to the decline of direct insulin action at several brain sites.
A ketogenic diet tends to reduce cravings and therefore seems to allow people to control their appetite, instead of their appetite controlling them. The combination of reduced cravings plus a fat-burning metabolism allows many people to lose a lot of fat, particularly if they had been insulin-resistant before. Furthermore, some of these ketone bodies are “lost” via the urine and the breath, which may contribute a little to the weight loss.
It usually takes around 3-6 months to be fully adapted to a ketogenic diet (faster if people engage in regular long-term fasting or fasting-mimicking diets). This adaptation mostly takes place at the epigenetic level in the form of up and downregulation of a number of genetic programs.
During the first 3-6 months, endogenous glucose requirements are higher than dietary intake can provide for, and during this time cortisol levels are elevated to ramp up the process of gluconeogenesis.
Interestingly, many people seem to feel best during this initial time when cortisol levels are through the roof (boundless energy; mental clarity; etc.). Even more interestingly, many people stop the ketogenic diet at the 3-6 month mark, a time when cortisol levels start to decline to more physiological levels.

Besides cortisol, a ketogenic diet also affects many other aspects of the endocrine system. Among other things, insulin stimulates the hepatic synthesis of IGF-1, the synthesis of leptin, and the conversion of the inactive thyroid hormone T4 to the active thyroid hormone T3. Furthermore, insulin regulates the expression of several hormone-binding proteins, altering the bioavailability of sex hormones (SHBG), thyroid hormones (TBG), cortisol (CBG), and IGF-1 (IGF1BP-3). If insulin levels fall, all of these processes are effected: IGF-1 levels decline, leptin levels decline, and T3 levels decline.
Insulin is closely related to insulin-like-growth-factor 1 (IGF-1), both of which are perhaps the major circulating growth factors in the human body. Whenever levels of IGF-1 and insulin are low, autophagy is disinhibited, which comes with its own set of changes, including mitochondrial recycling, the breakdown of faulty proteins, and a host of maintenance and repair processes.
Last but not least, insulin affects a variety of immune cells in several ways. The combination of an elevation in ketone bodies, low insulin levels, and various endocrine changes also results in a potent anti-inflammatory effect, reducing circulating levels of cytokines.
In summary, a ketogenic diet alters many aspects of human physiology, sometimes at the deepest possible level (gene expression).
According to Dr. Peter Attia, a ketogenic diet works especially well for three groups of people:
- Individuals who are metabolically unhealthy (i.e., insulin-resistant).
- Individuals who need to be cognitively sharp most of the time with little fluctuations in energy levels (e.g., knowledge workers).
- Ultra-endurance athletes
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Is it safe to be on a ketogenic diet long-term?
A ketogenic diet is an effective and efficient way to lose body fat (particularly visceral fat). Through this and other mechanisms (some of which were discussed above), a ketogenic diet is a fantastic way to reverse metabolic disease, including hepatic steatosis, dyslipidemia, and insulin resistance. Therefore, a ketogenic diet may help people to become metabolically healthy, which is associated with a number of longevity benefits.
However, is it also a “healthy” diet to be on after metabolic disease is mostly reversed (i.e., once people are lean and insulin-sensitive)? Unfortunately, we do not have a proper answer to this. It seems that some people fare on a ketogenic diet much better than others, perhaps in part related to genetic differences (particularly in genes of the PPAR family of transcription factors).

There is evidence that Inuit people (who are mostly on a ketogenic diet) seem to age quickly, however, epidemiological data always have severe limitations due to many potentially confounding third variables.
I personally have never met anyone who has benefitted from a ketogenic diet for more than a year or so, though two of my friends have gone back and forth multiple times (cycling in and out of ketosis happens more efficiently and effectively the more often one does it).
My gut feeling is that the ketogenic diet is not the safest diet to be on for the long term, in particular due to the numerous changes in endocrine systems, which (often) include an increase in area-under-curve cortisol levels, and a decrease in thyroid hormones, sex hormones, IGF-1, and leptin, all of which are necessary for proper physiological function, vitality, and brain health. Some of these hormones are discussed in more detail here. Furthermore, one should watch LDL/ApoB levels on a ketogenic diet, particularly if it is rich in saturated fats. Most people should probably use a microdose of a statin.
It seems that some people do indeed well on a strict ketogenic diet for extended periods of time (even after metabolic health is restored) without ever crashing their mental, metabolic, or hormonal health, but most people seem to eventually burn out. Reportedly, Joe Rogan has been on this diet for multiple years.
Unfortunately, when it comes to the promotion of different diets, one only hears about the cheerleaders making a lot of noise but not about the countless mute corpses in graves. What is more, many of the initial cheerleaders turn into corpses over time and then quiet down. So, informational supply about certain diets (e.g., keto, carnivore, intermittent fasting, vegan, etc.) is certainly biased.
Drugs that can make a ketogenic diet easier
There are two drugs that can make a ketogenic diet easier.
Acarbose
Acarbose is a glucosidase inhibitor that “converts” fast-spiking carbohydrates (starches) to low-glycemic carbohydrates. This flattens glucose and insulin excursions. Taken before a meal, this allows for an additional carbohydrate consumption of about 20g per day or so.
SGTL-2 inhibitors
SGLT-2 inhibitors cause a urinary loss of about 40-50g of glucose per day while someone is on a ketogenic diet (under normal non-ketogenic conditions, the urinary loss of glucose is about twice as high). This increases ketogenesis, beta-oxidation, and free-fatty acid liberation. Furthermore, one can (and has to) eat about 40-50g of glucose more than one would be able to otherwise. A friend has been on this combination for many months and is doing well.
DANGER: Combining SGLT-2 inhibitors with low-carb diets can cause euglycemic ketoacidosis, so regularly measuring blood ketone levels (or urinary) ketone levels is essential. Some people have landed in the ER from this combination because they were not watching ketone levels.
Related articles:
Sources & further info
- Podcast: Peter Attia & Jason Fung: Fasting as a potent antidote to obesity, insulin resistance, type 2 diabetes, and the many symptoms of metabolic illness
- Scientific article: Ketogenic Diets and Chronic Disease: Weighing the Benefits Against the Risks
- Opinion article: Ketogenic Diet Risks: Is Keto Worth It?
Disclaimer
The content available on this website is based on the author’s individual research, opinions, and personal experiences. It is intended solely for informational and entertainment purposes and does not constitute medical advice. The author does not endorse the use of supplements, pharmaceutical drugs, or hormones without the direct oversight of a qualified physician. People should never disregard professional medical advice or delay in seeking it because of something they have read on the internet.
