As discussed at length here, aging is a natural, spontaneous, and progressive process. It happens to everyone simply as a byproduct of being alive. However, it can be somewhat counteracted and delayed if I am just aggressive enough with it.
My objective is to strike a decent balance between longevity and vitality (as unfortunately, longevity and vitality are sometimes at odds – for example, caloric restriction). I personally do not care about maximum life span extension as this comes with quality of life tradeoffs I am not willing to make (e.g., living a sheltered life like Bryan Johnson (my take on him here), food obsession, etc.).
Said in other words, my objective is rather to live better than to live longer. If I can add a healthy decade or so to my life that would obviously be great too.
I employ a multitude of different strategies, each targeting one or more aspects of the aging process from a different angle. Each strategies employs a number of tactics.

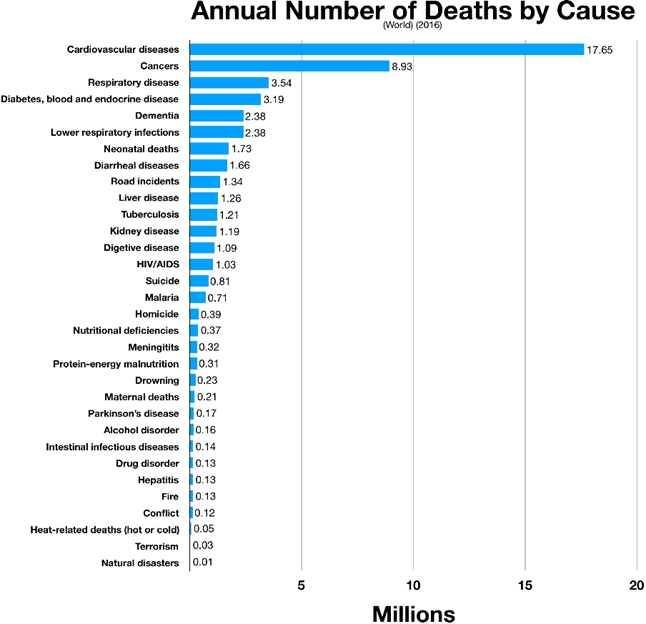
Half of solving the longevity equation comes down to delaying (or avoiding) chronic disease that currently kill about 80% of people in industrialized nations. Heart disease, cancer, and dementia. But in this article, I will cover much more than that.
Biology is chaos and due to the interconnectedness of bodily processes, there is necessarily some overlap between both strategies as well as tactics. For example, decreasing the activity of the mTOR pathway or keeping hormones at youthful levels will also help with counteracting inflammation, neurodegeneration, atherosclerosis, and deterioration of the musculoskeletal system.
A word on prevention
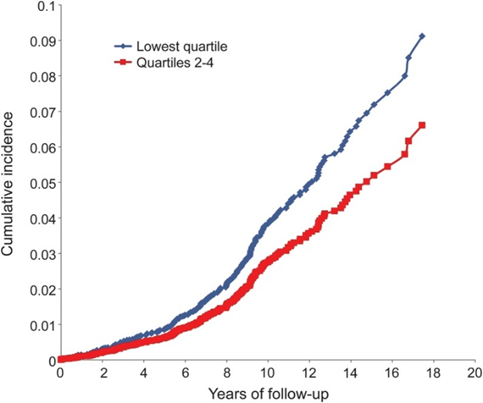
Once centenarians get cancer or heart disease they die at about the same rate as normal mortals. The difference is that they get these diseases one to two decades later. Therefore, if I want to live longer and better than average, I have to delay the onset of these chronic diseases.
Unfortunately, most healthcare systems in industrialized nations are geared towards helping people when there is a problem but not to prevent problems from occurring in the first place. As Dr. Peter Attia nicely put it, we live in a sick-care system but not so much a system of health care. In most modern sick care systems only about 1-3% of the total budget is spent on prevention.
Unlike with a car, I cannot replace the most crucial, shortest-half-life parts of my body such as the cardiovascular system, the nervous system, or the musculoskeletal system. Deleterious changes start accumulating long before they become evident. Therefore, ideally, treatment is started at the pre-pre-disease state.
Longevity is similar to investing. For maximum gains, I need to start as early as possible. Analogously, deleterious changes accumulate long (decades) before they become clinically evident and relevant. Therefore, longevity is largely about prevention. And almost by definition, prevention entails intervention.
However, many people I know are against intervening, which many assume is “bad” per se. Specifically, everything synthetic (pharmaceutical) has a connotation of “evil” (whereas “herbal” stuff usually does not).
Given intervention is done prudently, I argue that an individual who intervenes smartly is overall net better off than someone who does not use the fruits of modern medicine at all. I discuss this and other guiding principles in more detail here.
Everything has risks and side effects but so has not doing anything at all.
For example, let’s consider cardiovascular disease, which will roughly kill half of us. Atherosclerosis is a systemic condition of the arterial system and the first signs of it (fatty streaks) appear already in the first decade of life. It is unavoidable and progressive in everyone. Without intervention, I and everyone I know and love will get it eventually no matter how healthy they seem to be on the outside.
However, the rate of its progression can be slowed drastically by early intervention (prevention). And as so often, the risk of doing something must be weighed against the risk of doing nothing.
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part I: My Most Likely Cause Of Death – My Protocol for Fighting Atherosclerosis
Currently, about 50% of all deaths in industrialized nations are due to atherosclerosis, which is also the condition that kills most humans worldwide.
Preventing atherosclerosis is crucial for longevity
A lot of solving the longevity equation comes down to delaying the onset of atherosclerosis. The earlier risk factors are addressed, the better the chance to delay (or ideally, prevent) this disease and its consequences later in life. The consequences of atherosclerosis go far beyond cardiovascular events (such as heart attack, and stroke) and include a worsening blood supply to essentially all cells of the body.
The most well-known form of this is peripheral artery disease, which includes pain while walking (intermittent claudication) and poorly healing infections often leading to amputation. However, atherosclerosis progressively worsens blood supply even if the patient does not notice – which is often overlooked. The first signs are sometimes erectile dysfunction (worsening of blood supply to the penis) and even dementia (worsening of the blood supply to the brain), however, ultimately, blood supply is worsened to every cell, tissue, and organ because atherosclerosis is a systemic condition of the arterial system.
Unless humanity is able to fundamentally change how lipoproteins interface with the endothelium, this disease will progress in everyone, although the rate of progress can be vastly modified. The earlier the intervention, the better, as the longer my artery walls are exposed to atherogenic particles and other risk factors, the more likely they are to incur damage.
How atherosclerosis develops
Atherosclerosis (“cardiovascular disease”) is a time-course disease, which is why age is such a strong predictor of risk. The longer arterial walls are exposed to insults and atherogenic particles (lipoproteins) the more damage they will incur.
Atherosclerosis is fundamentally a progressive narrowing and degeneration of blood vessels. This disease starts already in the first decade of life and eventually kills people via heart attack, heart failure, or stroke.
There are multiple drivers that can lead to the development of atherosclerosis. The most widely known are high blood lipids (dyslipidemia), high blood pressure (hypertension), high blood sugar (hyperglycemia), and smoking. These so-called “Framingham factors” factors (excluding smoking) are often a direct consequence of metabolic disease.
Some of the contributing factors are high levels of Lp(a), low-level inflammation, hypercortisolemia, or low levels of thyroid hormones, sex hormones, or growth hormone/IGF-1. Furthermore, there are a large variety of genes that are thought to influence atherogenesis in one way or another. In fact, genetics presumably play a huge role. Some people live well into their 90s with sky-high levels of ApoB or lp(a) whereas others have CV-events early, without any obvious risk factors.
As always, biology is messy: Multiple things occur in parallel, and everyone has a unique constellation of protective and predisposing factors.

How does atherosclerosis happen?
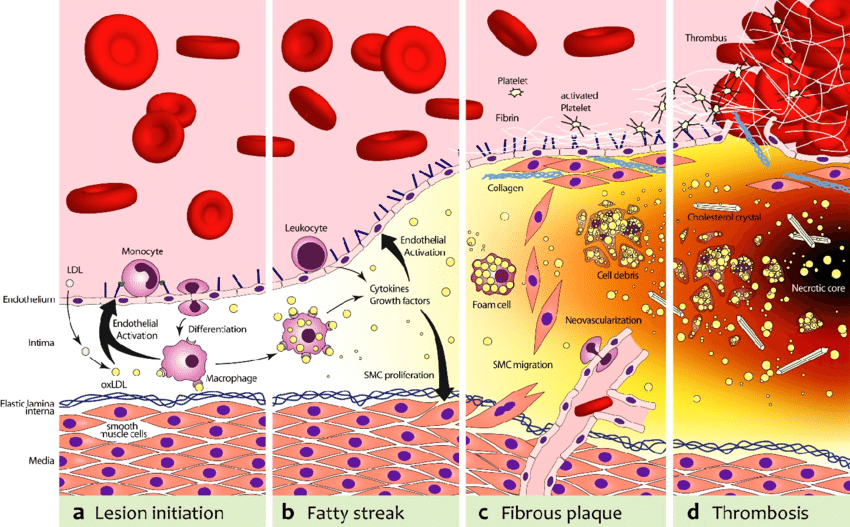
The first step in atherosclerosis is injury to the innermost lining of blood vessels (endothelium) by a variety of mechanisms, including smoking, hypertension, or hyperglycemia. The ensuing inflammatory reaction then inflames and thickens the endothelium (“intima thickening”).
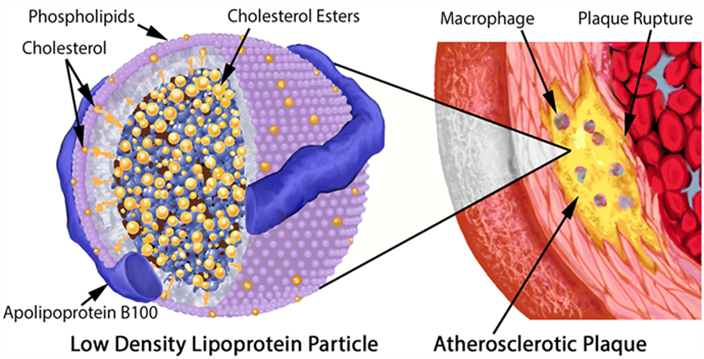
At the same time, lipoproteins (essentially LDL particles) get into the inflamed vascular wall. If these LDL particles come in contact with the pre-damaged vascular wall, they are oxidized. (Technically, LDL particles by themselves are thought to be harmless, and the ultimate atherogenic compounds are oxidized molecules contained within them.) These oxidized LDL particles are then engulfed by macrophages. Macrophages die and cause even more inflammation. Fibrosis (scar tissue) develops and narrows the artery.
In addition to the narrowing, depending on how bad the inflammatory environment is, a lipid-laden cap starts to cover the lesion (“plaque”), which renders it vulnerable to rupture.
Upon rupture, blood clotting can occlude the artery and cause an infarction (e.g., if a cardiac artery is occluded this is called a heart attack). Alternatively, the ruptured lipid-laden mess can be carried away and occlude an artery somewhere downstream (e.g., a plaque that forms in the heart atria dislodges and is transported into the circulatory system of the brain causing a stroke.)
Risk factors
Unfortunately, there are many risk factors at play:
- A large number of genes
- The number of lipoproteins cruising through the bloodstream at any given time (measured by ApoB – the protein found on the surface of atherogenic lipoproteins mostly responsible for driving atherosclerosis)
- Lp(a) particles (LDL particles with an apolipoprotein(a) attached to them)
- Anything that causes injury to the endothelium. First and foremost, high blood pressure, high blood sugar, and smoking.
- A variety of hormones, all of which have a variety of direct and indirect effects on endothelial health, metabolic health, and repair capabilities.
- Levels of inflammation
There are many other “obscure” genetic factors at play that are poorly understood
Atherosclerosis can run in families even when there’s no obvious cause. They don’t have FH (familial hypercholesterolemia) or high Lp(a). Their apoB is around the 50th percentile of the population. Yet, they are disproportionately afflicted at a young age – meaning, a heart attack in their fourties.
Similarly, there are tons of patients who’ve made it to their 90s with high apoB levels (and even high lp(a) levels) that don’t have obvious atherosclerotic disease. Nonetheless, the evidence is quite clear and we understand atherosclerosis better than most other multifactorial diseases in medicine. Just because some people can get away with chain-smoking for life does not mean that chain-smoking is not the root cause of most cases of COPD or lung cancer.
Tactics I follow to delay atherosclerosis
- Blood pressure
- Managing lipoproteins
- Hormone optimization
- Metabolic health
- Avoiding spikes in blood sugar
- Keeping insulin levels fairly low
- Maintaining a low body fat percentage
- Optimizing endothelial health
- Lowering inflammation
- Exercise
- Lp(a)
- TMAO & homocysteine
Keeping an eye on blood pressure
Out of all the Framingham factors, hypertension is the deadliest. It is thought that hypertension kills about 9 Mio. people per year, which is perhaps 10-15% of all deaths. For every 10mmHg reduction in RR, the risk for cardiovascular events drops by 45% (data derived from Mendelian randomization).
Blood pressure leads to atherosclerosis through injury to the intima, the innermost lining of arteries. This then leads to intima thickening and atherosclerotic progression (as explained above).
High blood pressure runs in my family and I have a couple of (potentially) deleterious single nucleotide polymorphisms (SNPs) in my renin-angiotensin-aldosterone system (RAAS). However, fortunately, my blood pressure is usually around 120/65 mmHg.
Obviously, weight control and exercise, both of which have independent effects on blood pressure, should be taken care first. However, blood pressure has strong genetic underpinnings.
If I were to choose a drug for hypertension, I would choose telmisartan over other antihypertensives. Telmisartan is an angiotensin receptor blocker (ARB). It has a long half-life and beneficial effects on metabolic health because of its off-target effects on PPAR-delta (a transcription factor involved in fatty acid metabolism). PPAR-delta modulators are banned by WADA because of their effects on endurance capacity. Unfortunately, most doctors choose ramipril or valsartan as their first-line antihypertensive – mostly because of historical reasons.
Of note, automatic RR-measuring devices often overestimate the systolic pressure by 5-10mmHg.
Managing lipoproteins
The lower lipoproteins are (particularly LDL levels) the better. Kids have very low LDL levels (roughly 20-30mg/dl) and they do not suffer any adverse effects. Similar things hold true for people with a mutation in the PCSK9 gene (hypofunction). They have very low LDL levels (sometimes below 20mg/dl) but do not have any increased risk of dementia, depression, or any other disease – but they are virtually free of atherosclerosis (including heart attacks and many forms of stroke).
As explained above, lipoproteins (essentially LDL particles) get into the inflamed vascular wall where they are oxidized. These oxidized LDL particles are then engulfed by macrophages. Macrophages die and cause even more inflammation. Fibrosis (scar tissue) develops and narrows the artery, often with a lipid-laden cap.
Consequently, lipoprotein particles are the causative agent of atherosclerosis (“necessary but not sufficient”). For every 40mg/dl reduction in LDL, the risk of CV events drops by 55% (Mendelian randomization data). Similarly, a reduction of 25mg/dl LDL + a reduction of 5mmHg RR reduces the risk by 55% as well.
Atherogenesis is sterol-mediated, but sterols are trafficked within ApoB-containing lipoproteins. In other words, it is the number of ApoB particles that is most relevant. Hence, ApoB is the blood marker one should test for.
Since I started doing regular blood tests about a decade ago, my ApoB levels are consistently below the clinical reference range (ApoB between 35-50mg/dL and my HDL at around 70mg/dL – though it is becoming clear that HDL is a mostly worthless marker). Interestingly, no matter what diet I follow (e.g., keto, intermittent fasting, high-carb diet) my ApoB levels do not seem to change much.
This means that I am naturally in the top 2.5% of the population. Initially, I thought they were that low because of all the drugs and supplements I experiment with but I eventually found out that my brother, who barely takes anything, also has ultra-low levels. So I guess I am blessed in this regard (I discuss my genetic test results in more detail here). However, fish oil and rapamycin may drive them even lower.
If my ApoB levels were an issue, I would not even bother with lifestyle interventions (which do not do much for lipids anyway) but would immediately jump to 2.5mg of rosuvastatin (half of the lowest clinical dose) plus 10mg of ezetimibe.
Rosuvastatin is a statin (blocking HMG-CoA-Reductase – one of the key enzymes in the synthesis of cholesterol) and at low doses, it is mostly liver-targeted. Furthermore, it is very hydrophilic compared to other statins and the lack of lipophilicity keeps it mostly outside the blood-brain barrier.
2.5mg of rosuvastatin is known to knock down ApoB levels by about 40%. 40mg of the drug only achieves about 55% despite being 16x the dosage (consequently with more systemic effects). Unfortunately, in the clinic, I see 40mg of rosuvastatin given out all the time.
A friend who is statin-intolerant has capsules containing 1mg of rosuvastatin compounded for himself, which he can take without side effects (for reference: the lowest clinical dose is usually 5mg).
In my opinion, from an angle of primary prevention, there is little need for clinical doses, which come with a greater incidence of adverse effects but do not lower LDL by much more anyway. Statins are also thought to lower inflammation, as evidenced by a decrease in CRP levels upon starting statin treatment.
Three other drugs commonly used for lowering LDL particle count are:
- Ezetimibe – a blocker of the absorption of endogenous cholesterol. Can be added to all the other lipid-lowering agents. Mostly devoid of side effects. I personally use 5mg of ezetimibe per day.
- For statin-intolerant people there is bempedoic acid. An amazing drug, though unfortunately quite expensive and/or hard to get approved for.
- Then there are the mighty (but even more expensive) PCSK9 inhibitors, blockers of an enzyme responsible for the recycling of LDL receptors. They are also known to lower lp(a) levels by about 30%. Recently, inclisiran has been added to the arsenal. Inclisiran is a siRNA that needs to be injected every six months. It also inhibits the synthesis of PCSK-9 but by a different mechanism than PCSK9 inhibitors.
There are benefits of starting lipid-lowering therapies and blood pressure-lowering therapies as early as possible as the progression of atherosclerosis is both a function of apoB concentration & blood pressure and duration of exposure. This is similar to smoking, where the damage is cumulative and measured in “pack-years” (1 pack-year being 1 year of smoking one pack per day).
Of note, HDL (also known as “good” cholesterol) is no longer considered informative to use and a higher HDL is associated with better lipd levels but it may not have any protective effect by itself.
Hormone optimization
Thyroid hormones, growth hormone/IGF-1, and sex hormones are all quite highly anti-atherogenic. I make sure that all of these are “good” (youthful levels). Once my other hormones are going to decline, I will seriously consider replacing them. Hormones are discussed here.
For women, atherosclerosis vastly accelerates after menopause. Before menopause, E2 acts as a potent protective factor. On average, women still have a delay in atherosclerosis by about 10y.
Metabolic health
Metabolic health is discussed in depth in another article. In short, my strategies include keeping body fat levels low, endurance exercise, HIIT, maintaining a decent amount of muscle mass, rapamycin, optimizing hormones, keeping insulin sensitivity high year-round, diet, sleep, and metabolic drugs.
Avoiding spikes in blood glucose
I try my best to avoid spikes in blood glucose whenever I am eating by myself – I do not care about spiking blood sugar during social occasions.
Whenever levels of blood glucose are high, glucose covalently bonds to certain amino acids on proteins (Maillard reaction) leading to the formation of advanced glycogen end products (AGEs). These AGEs are phagocytized by macrophages through the RAGE-receptor (among others), which stimulates inflammation.
Furthermore, due to the covalent crosslinking, protein structure (and therefore function) is compromised. Both of these (AGEs; protein function) play a prominent role in micro-vascular disease, which is among the leading causes of blindness, renal failure, and neurodegeneration. Furthermore, it accelerates macrovascular disease (atherosclerosis).
Keeping insulin levels fairly low
While insulin will lower blood glucose (which is beneficial for micro-vascular disease), insulin itself promotes the development of macro-vascular disease (atherosclerosis), which includes coronary vascular disease (leading to heart attack), cerebrovascular disease (leading to stroke), and peripheral vascular disease (leading to amputation).
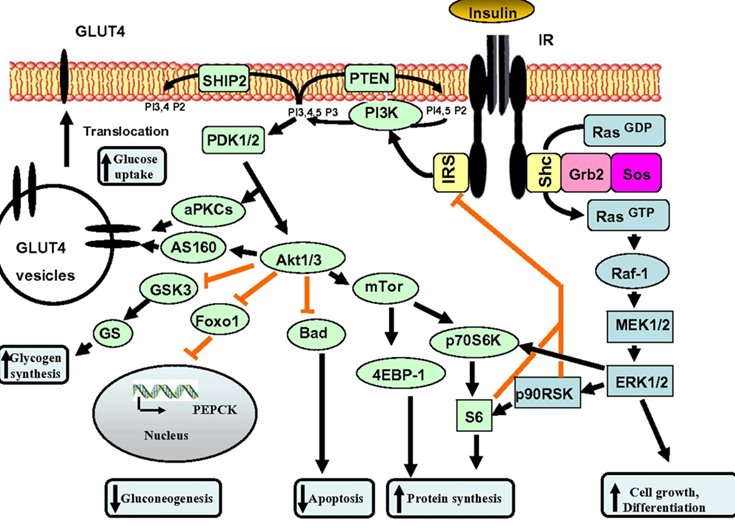
Unfortunately, few physicians realize that controlling blood glucose (HBA1C) is only half of the job, and the other half is about keeping area-under-curve levels of insulin low. A lot of this comes down to insulin being a potent growth factor, and “insulin all of the time” equals “growth all of the time”, which is driving pretty much every chronic disease, including atherosclerosis.
The two best ways to keep area-under-curve insulin levels low without having to fast is to reduce consumption of fast-spiking carbohydrates (“bad carbs” – I usually only consume “bad carbs” after exercise, a time when my liver and muscle are primed to mop up excess glucose to resynthesize glycogen) and to keep insulin sensitivity high. I discuss the strategies I use to keep insulin sensitivity high here.
Maintaining low body fat percentage
I try to keep my body fat low. This keeps me insulin-sensitive and lowers lipid levels in my bloodstream because adipocytes are primed to absorb them. A better lipid profile (essentially lower levels of ApoB and triglycerides) means less atherosclerosis.
Things that help me with this are a hefty dose of exercise, adhering to a couple of dietary principles, optimizing my hormones, and a very low dose metreleptin and on-and-off use of semaglutide .
Optimizing endothelial health
Similar to how it is hard to define what “metabolic health” is, it is hard to define what endothelial health is. I try to keep my endothelium healthy by optimizing hormones, rapamycin, my past use of senolytics (?), antioxidants, and exercise.
I experimented on and off with a very low dose of tadalafil (1mg/d) to increase cardiovascular health and for its putative anti-inflammatory benefits. Tadalafil is a PDE5 inhibitor, frequently used for erectile dysfunction.
The idea is that the boost in nitric oxide signaling will deliver a lot of cardiovascular benefits, which is also presumably one reason why eating vegetables is associated with better cardiovascular health (vegetables contain a lot of nitrates, some of which convert into nitric oxide).
Furthermore, PDE5 inhibitors have also been shown to have anti-inflammatory, antioxidant, antiproliferative, and metabolic-health-enhancing properties in several experiments.
However, while they likely did improve my endothelial health (at least as judged by my forearm vascularity as a pathetic proxy), my most recent experiment led to a sudden onset of tinnitus within 30min of taking 1mg of tadalafil.
Given that PDE5 inhibitors are highly associated with new-onset tinnitus and weakly associated with hearing damage, I decided to stop tadalafil for good, as even a small chance of sensory impairment (and therefore loss of quality of life) is not worth a small cardiovascular benefit in an otherwise healthy young person. Fortunately, the tinnitus went away after 48 hours.
“Fun” fact: Hugh Hefner, the founder of Playboy, blames his almost complete hearing loss on the overuse of sildenafil (Viagra), and there is reason to believe that there is a causal correlation.
Lp(a)
As I was working in cardiac and vascular surgery, there were lots and lots of amputations happening all the time. When patients are lucky, it is just a toe. But most of the time, it is the whole foot or lower leg.
Most of the patients have a combination of long-standing poorly controlled hyperglycemia and full-time smoking. The endothelial inflammation and the neuropathy lead to non-recognized and poorly healing infections which may eventually turn into sepsis – hence the preemptive amputation.
One time, however, there was a lower leg amputation on a guy who is barely 45 years old. He was not even overweight and did not have diabetes. While he was a smoker, the primary reason for losing his lower leg at such a young age was elevated levels of lp(a).
In his case, the elevated levels of lp(a) led to early atherosclerotic degeneration of blood vessels, including manifest peripheral artery disease. A small injury led to a poorly healing infection, which turned into the requirement for amputation.
Lp(a) is an insidious particle that is much more important than commonly appreciated. Through pro-inflammatory mechanisms, it drives the development of atherosclerosis. Patients with high levels of lp(a) often have major cardiovascular events (e.g., heart attack, stroke) and peripheral artery disease at a young age.
About 10% of people have genetically elevated Lp(a), which may have conferred an advantage in blood clothing in the past. Again, evolution favored something that is awesome for the young at the expense of the old. There is quite little one can do to lower lp(a) beyond turning to cutting-edge pharmacology.
Fortunately, my Lp(a) is naturally low (around 10mg/dl). If my Lp(a) were high, I would turn to PCSK9 inhibitors to lower it. There is also an antisense drug on the way that will hopefully be available for clinical use soon (pelacarsen). There are also various gene therapies currently in various phases of clinical development.
TMAO & homocysteine
Next to lp(a), also TMAO & homocysteine are associated with atherosclerotic risk.
TMAO is a small amine molecule found in many marine species to help preserve protein stability at great depths. It is thought that in mammals, TMAO activates macrophages, thereby leading to inflammation, which then promotes atherogenesis but the exact mechanism is still unclear.
In humans, TMAO can be produced by certain gut microbes in response to carnitine and cholines (e.g., alpha-GPC, phosphatidylcholine). I supplement with both of them. Unfortunately, thus far, I have not been able to find a laboratory that measures TMAO levels.
Homocysteine is an amino acid produced as a byproduct during the metabolism of methionine (an essential amino acid). It is thought that homocysteine is an autoreactive molecule that produces oxidative stress. High levels of homocysteine seem to be correlated with cardiovascular disease. Fortunately, my homocysteine levels always came back low when I tested for them. TMG, a supplement I take, is known to lower homocysteine levels as are methylated B vitamins.
However, in comparison to the importance of blood pressure & ApoB & lp(a), TMAO & homocysteine are barely worth mentioning!
Lowering inflammation
Discussed extensively here.
Exercise
I do about 1 hour of exercise per day. Usually, I alternate between the gym and steady-state cardio (so-called “zone II exercise”). Twice per week I also work on my mobility. I discuss my exercise regimen in more detail here.
Sources & further information
- Opinion article: Peter Attia – When does heart disease begin (and what this tells us about prevention)?
- Podcast: Peter Attia & Ron Krauss – A deep dive into heart disease
- Website: Ototoxic Adverse Drug Reactions: A Disproportionality Analysis Using the Italian Spontaneous Reporting Database
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part II: My Protocol for Fighting Cancer – The Ultimate Frontier
Cancer is called “the emperor of disease” for a reason. To some degree, no matter what I do, my likelihood of developing cancer at some point is between 30-60%. Out of all of the things that are known to kill humans, cancer is perhaps the hardest to figure out, in part, because it also depends on bad luck.
Furthermore, cancer is not one disease but is a collection of hundreds of different sub-diseases because tumorigenesis in different tissues is quite different, requiring different treatment approaches.
However, there are a number of things I can do now to vastly decrease my chances of getting an advanced form of it – more on that in the second part of the article.
How does cancer arise?
There are a variety of causal associations between cancer and genetics, pathogens, or environmental exposure. However, most cancers are due to random mutations because of “bad luck”.
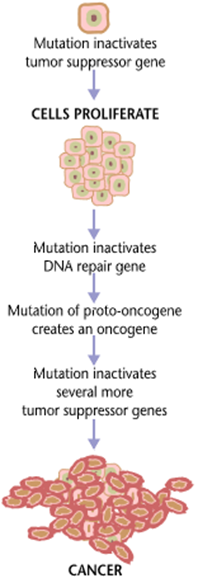
Cancer is fundamentally caused by genetic mutations, which cause a cell to behave in ways it should not. In the case of cancer, this means “growing and proliferating” at the expense of other cells. Cancer is like having a foreign parasite inside your own body, with the exception that the parasite is not “foreign” – at least not initially.
Because somebody else said it better than I ever could, here is what oncologist Keith Flathery said about the pathogenesis of cancer:
“The combination of mutations has to be dialed in the right sequence just like when you’re opening your gym locker, or you don’t get cancer. You’ve got to get your tumor suppressor early and in the right order before your activated oncogene comes along.
You’re randomly spinning a lock. You’re picking up lots of past permutations, not just true drivers, so these incidental mutations happen here and there. As that’s happening, you finally click important components of the program. Along the way, some of those are actually mutations that are seen as foreign by the immune system.
If they’re too far out there, then they’re gone. It has to be a right kind of genetic alteration that will give the cell what it needs to be able to proliferate abnormally, be able to sustain a lot of DNA damage as it accumulates, and not commit suicide as a consequence, and be able to handle all of the other adverse features and filters of a tumor micro-environment.
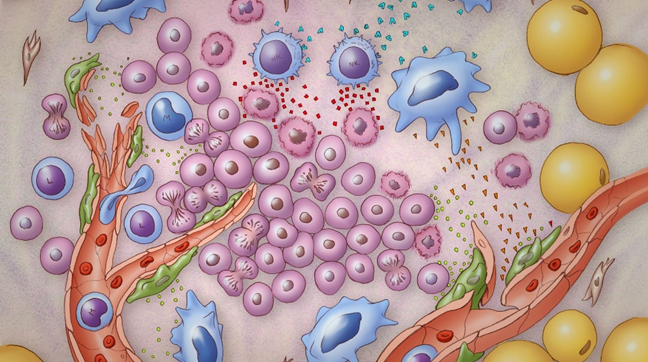
By the time the tumor mass is clinically relevant, there is a mass of about one billion cells. They have had to deal with an incredible array of defense mechanisms and cellular brakes. Tumor cells are evolutionary warriors by this point and using our chemotherapies is like poking them with a stick. Some subpopulations of those cancer cells will slow down and die with conventional chemotherapy, but many of them are pre-wired. They were hardy to begin with.
They got there through a hard-earned evolution under selective pressure of the immune system and adverse metabolic environment, and they used all of the tricks up their sleeve to reprogram themselves to be able to survive in these harsh environments. You throw in another harsh environment reagent in the form of chemo, not surprisingly, these things were already basically hardwired to be able to survive yet another insult.”
For people interested in cancer biology and treatment, I recommend the full podcast.
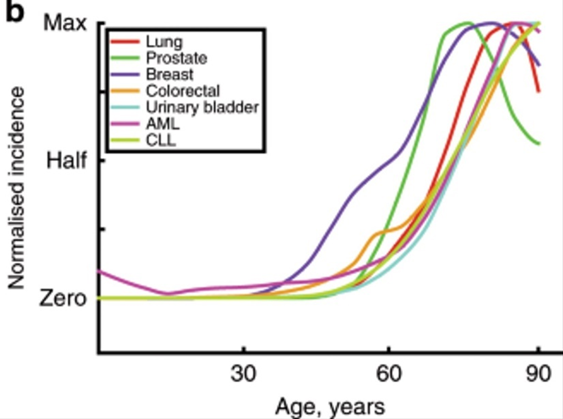
There are a number of reasons why my risk of developing cancer increases non-linearly as I get older:
- As I get older, my genome is more susceptible to injury (e.g., DNA-repair machinery and other cellular housekeeping functions degenerate due to aging)
- Mutational load is a function of time. The older I am, the more of these genetic “hits” (cancer-causing mutations) I have already accumulated, and the fewer additional hits are needed for cancer to develop.
My immune system will gradually lose some of its steam (“immunosenescence”), and the ability with which it detects and dismantles early cancer cells progressively declines.
By the time cancer becomes clinically relevant has become sufficiently advanced, the lucky subclones will just laughingly mutate their way around everything we throw at them.
Currently, close to 100% of the time, metastatic cancer (of a solid organ) is deadly (with very few exceptions), and survival can only be prolonged slightly (a few weeks to months for tens of thousands of dollars). Hence, once there is metastasis, there is little that can be done. So, the most efficient & effective way to go about it is undoubtedly prevention.
Cancer prevention
Currently, there are two things people can do to increase the odds of not dying from cancer.
Step 1: Trying not to get cancer
We know of a handful of things that undoubtedly increase cancer risk. These include smoking (thought to be responsible for about 30% of cancers), obesity (especially the associated hyperinsulinemia), exposure to certain carcinogens (hint: it is not red meat), and a number of infections (e.g., HPV, HCV, HBV).
Step 2: Looking for cancer early
When it comes to cancer, early detection is the name of the game because it is about the only thing that has worked thus far (aside from a couple of non-universal breakthroughs for a specific subset of cancers). Currently, this includes keeping an eye on a host of plasmatic tumor markers, and occasionally performing screening procedures (such as colonoscopy, mammography, measuring PSA levels, and full-body MRIs).
In the (near?) future, progress in the x-omics section (e.g., genomics, transcriptomics, proteomics, metabolomics) will presumably revolutionize cancer detection because they may yield valuable biomarkers. For example, “liquid biopsies” will be fine-tuned with machine-learning approaches (“artificial intelligence”). These biomarkers will then be layered with cutting-edge scanning and imaging techniques.
By layering multiple of these technologies on top of each other, perhaps with AI algorithms, we will hopefully get to a point where we can catch most cancers already in the early stages, long before they are ever in a position to leave the primary cancer site. Instead of spending 5% on prevention and early detection, we should spend 50% on it.
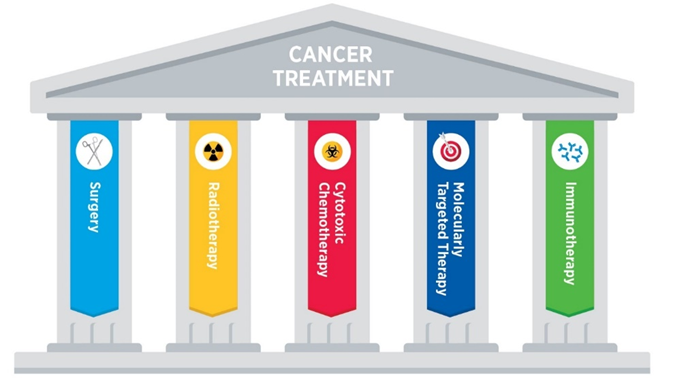
Once advanced cancer is there, it is probably best to go after multiple avenues simultaneously, including surgery, radiation, chemotherapy, immunotherapies, metabolic therapies, epigenetic regulators, and growth factor receptor modulators.
Because this section is already quite long, I discuss liquid biopsies, immunotherapies, and potential improvement in the way current chemotherapy is done, in more detail here: Exciting New Avenues to Revolutionize Cancer Treatment
Tactics I follow aimed at preventing cancer
- Measuring stuff
- Rapamycin
- Full-body MRIs
- Keeping my insulin sensitivity high
- Vaccinations
Currently, there is nothing I do specifically to prevent cancer. However, lowering inflammation, lowering the activity of the mTOR pathway, and optimizing my metabolic health (especially keeping my AUC levels of insulin low) likely goes a long way. All of these are extensively discussed in later sections.
Measuring stuff
Once per year, I measure an extensive panel of cancer blood markers – which though are unfortunately neither very specific nor very sensitive. But it does not cost me anything and I have no side effects from a blood draw. Fortunately, I am still young enough so that by the time I will be in the midst of my thirties, there may be better measurements commercially available (e.g., proteomics, metabolomics).
If things progress as they currently stand, it is quite likely that in 10 years from now (barring human extinction or civilizational collapse) I can just perform a blood draw which then tells me the likelihood of having a cancer in a specific tissue – the tissues with high likelihood I can then run further diagnostics on.
Rapamycin
I take 3mg of rapamycin once per week. Rapamycin has potent antiproliferative effects and therefore has presumably widespread anti-cancer benefits. I discuss rapamycin in much more detail here.
Full-body MRIs
Thus far, I have performed two full-body MRIs, and I plan to keep doing these every 3-5 years. I only did my first one with gadolinium because of also checking for aneurysms but all the future ones will be without contrasting agents given that they are known to accumulate in the brain.
Insulin sensitivity
About 1/3 of cancers are caused by “diet”, most of which is due to hyperinsulinemia. I make sure that my insulin sensitivity is high year-round. I discuss my strategies here.
Vaccinations
Vaccination against HPV not only protects against sexually transmitted condylomata but also against a number of cancers. HPV worse for women (e.g., cervical cancer) than men but nonetheless, HPV vaccination protects against a number of other cancers including cervical cancer, penile cancer, anal cancer, and oropharyngeal cancers – in all these tissues as a rule of thumb about 70% are HPV related.
Vaccination against HBV protects against HBV-induced hepatocellular carcinoma. In fact, about 700.000 people per year (!) die worldwide every year because of hepatitis B virus induced hepatocellular carcinoma (liver cancer).
Sources & further information
- Scientific article: The hallmarks of cancer – New dimensions
- Scientific article: Cancer and Aging: Two Tightly Interconnected Biological Processes
- Podcast: Peter Attia & Keith Flaherty: Deep dive into cancer— History of oncology, novel approaches to treatment, and the exciting and hopeful future
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part III: My Protocol for Fighting Dementia – What I Do To Keep Brain Health Optimal
My nervous system is the basis of everything I think, do, and feel. To me, it is of utmost priority that it stays in the best possible shape. However, just as any other cell, tissue, or organ, the brain deteriorates over time.
Humans usually peak around 20-25 years in terms of mental capabilities. From then on, it goes downhill for everybody. This includes gradually progressing compromised blood flow, reduced neurogenesis, and lower neuron health. There is no way to completely prevent this.
Furthermore, as I age, I will progressively and stochastically lose neurons (in addition, to a decline in neuron function), which is likely part of the reason people’s cognitive abilities progressively decline as they age. For the most part, neurons in the CNS are postmitotic cells (i.e., they cannot divide) and neurogenesis is limited to a few select sites.
Even if we could replace neurons, newborn neurons are mostly incapable of replicating the functional connectivity of a dying neuron, particularly the sometimes macroscopic length of the axon. Thus, in contrast to other cells of most organ systems, neurons cannot be replaced. I discuss this in more detail here: Intelligence Enhancement
Dementia 101
While some form of neurodegeneration (e.g., loss of neuron number and function) happens to everyone, not everyone will get diagnosed with dementia.
It is only called “dementia” when neurodegeneration has progressed to the point that mental capabilities become so bad that they severely impair daily living. However, by that point, mental function has usually already been declining for a couple of decades.
“Dementia” is a catch-all term that includes a couple of neurodegenerative diseases that often present in a mix & match form. Most of these diseases are characterized by the aggregation of one or more faulty proteins. For example:
- Alzheimer’s dementia: The brains of people with Alzheimer’s disease are full of extracellular plaques of beta-amyloid and intracellular aggregates of tau protein, though it is still unsure whether these protein aggregates are a cause or a consequence of the disease. There are probably multiple different paths that lead to Alzheimer’s. These include vascular, metabolic, inflammatory, metabolic, toxic, and genetic “versions” of the disease – and it is not even clear whether different “kinds” of Alzheimer’s represent the same “disease”. About 80% of all dementia forms are Alzheimer’s dementia.
- Frontotemporal dementia: Frontotemporal dementia is associated with aggregates of another set of proteins (tau, TDP-43, FUS), particularly in frontal and temporal regions.
- Others: Faulty proteins also accumulate in Lewy body dementia, and Parkinson’s disease.
While we have identified a handful of clear-cut risk factors, nobody exactly knows why in certain people a certain subset of neurons degenerates. Furthermore, nobody knows whether the protein aggregates are a cause, byproduct, or both.
Tactics I follow to preserve brain health
- Having no ApoE4 allele
- Putting a prime on exercise
- Optimizing metabolic health
- Preventing atherosclerosis
- Optimizing hormones
- Lowering inflammation
- Fish oil
- Preventing nutrient deficiencies
- Senolytics
- MAO-B inhibition
- Optimizing sleep
- “Brain stimulation”
Genetics
By far the biggest aspect of avoiding dementia is choosing the right parents. My grandfather recently died at 96 years old. He died with no discernible dementia and his memory was as sharp as ever despite having been strongly hearing-impaired for over a decade – hearing impairment is highly associated with developing dementia. Furthermore, he had been using lorazepam for sleep for many years. Lorazepam is a strong benzodiazepine, which is strongly associated with cognitive decline.
Having no ApoE4 allele
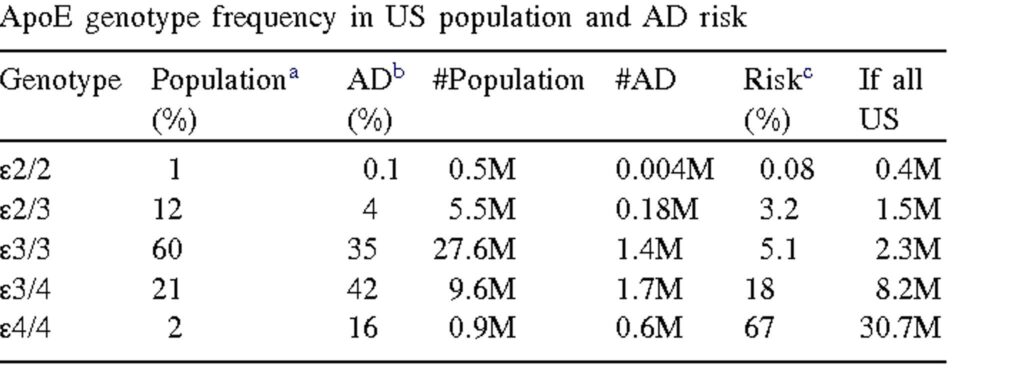
ApoE4 is a protein found on lipoproteins, and the ApoE4 version is associated with an increased risk for dementia, in particular Alzheimer’s, and cardiovascular disease. ApoE is the principal cholesterol carrier in the brain and seems to be involved in cholesterol transportation from astrocytes to neurons.
About 25% of the population has one ApoE4 allele. Having one ApoE4 allele roughly triples one’s risk for developing AD. One’s ApoE genotype is strongly correlated with the lifetime risk of Alzheimer’s dementia.
For example, carriers of the E4/E4 genotype, about 2% of the population, have a lifetime risk of about 70%, whereas carriers of the E2/E2 genotype are pretty much protected from developing AD. Nonetheless, these genes are not deterministic and about 1/3 of people with the E4/E4 genotype do not seem to develop AD. Likewise, a very small fraction of people with the E2/E2 genotype do develop AD.
I personally carry the E3/E3 genotype, which 60% of the population have.
Putting a prime on exercise
I do about 1 hour of exercise per day.
As of yet, exercise is probably the most potent way I have for staving off neurocognitive decline. After all, next to regulating homeostasis, the brain is all about movement (behavior) anyway.
For me, the primary reason I do lots of exercise is not related to metabolic health or looks, but rather to elevate brain function (e.g., executive functions) and to promote long-term brain health.
I discuss the benefits of exercise on vitality and brain health in more detail here.
All three kinds of exercise have (partially) independent benefits.
- Low-intensity cardio (zone II): it particularly increases mitochondrial and metabolic health.
- High-intensity cardio: It particularly boosts the release of a host of growth factors such as VEGF and BDNF, and increases the genetic expression of antioxidant systems.
- Resistance exercise: It particularly improves insulin sensitivity, IGF-1, and myokine signaling.
Optimizing metabolic health
Optimizing metabolic health is crucial to preserve brain function, presumably through a plethora of interconnected mechanisms. Furthermore, it counteracts the development of cardiovascular disease (discussed next).
I discuss metabolic health here: The Seven Pillars of Metabolic Health
Preventing atherosclerosis
There is a saying among doctors that “Everything that is good for the heart, is good for the brain.” In fact, there is a linear relationship between the media thickness of the internal carotid artery and the decline in cognitive function or said in another way, there is a strong relationship between atherosclerosis and neurodegeneration.
Furthermore, next to dementias associated with the accumulation of faulty proteins, there are so-called “vascular dementias”, which are caused by insufficient blood supply (oxygen, glucose, nutrients, hormones) and micro-strokes (the latter of which often go unnoticed), both of which lead to neuron death.
The tactics I follow to prevent or delay atherosclerosis are discussed here: My Most Likely Cause Of Death – My Protocol for Fighting Atherosclerosis
Optimizing hormones
I keep an eye on my hormones. Multiple hormones have beneficial effects on the brain as they also function as growth factors and metabolic modulators. These include T3, IGF1, GH, estradiol, and leptin. Neurosteroids (converted in part from other steroids) may also play a role however, their clinical importance is currently uncertain.
Conversely, if one of the major hormones is deficient, this will accelerate brain aging. At the time of this writing, I supplement with a low dose of leptin and I am on my TRT Lite protocol.
I am specifically pleased that my IGF-1 levels are naturally slightly above the reference range. IGF-1 is one of the major systemic growth factors. While it slightly increased the risk of cancer, it reduces the risk of cardiovascular disease, metabolic disease, and specifically neurodegenerative disease. In fact, low levels of IGF-1 are strongly associated with brain atrophy and dementia.
Furthermore, higher GH & IGF-1 are associated with more vitality as they improve energy levels, mood, overall health, and muscle mass, all of which are essential to living a brain-health-conducive lifestyle.
Growth hormone and leptin in particular increase REM sleep, which is crucial for brain plasticity.
Lowering inflammation
Low-level inflammation is toxic to brain integrity through a variety of mechanisms. Tactics aimed at reducing inflammation are discussed here: Burning to Death – My Protocol for Lowering Inflammation
Fish oil
I take 2g of EPA/DHA per day. Simplistically speaking, EPA is a structural building block of neurons, while DHA is a precursor to anti-inflammatory prostaglandins. We do not really know which of these is more important so it is probably best to include both.
40%-50% of the brain is composed of PUFA. DHA is the main building block and most common in the brain. EPA is less abundant but may have more anti-inflammatory properties compared to DHA. Therefore, elevating O3-fatty acid intake may be somewhat protective against dementia.
Preventing nutrient deficiencies
I make sure I have no mineral or nutrient deficiencies by occasionally measuring some of these in my blood.
I take a load of supplements – discussed here: Supplements I Take
Senolytics
It was found that senolytics reduce senescent oligodendrocyte progenitor cells, reduce beta-amyloid plaque in the hippocampus, reduce neuroinflammation, and improve memory and learning in mice. In animal models, rapamycin acted on the early stage of dementia but senolytics were found to act on the later stages, ameliorating already established disease. Unfortunately, there is currently no human data. I discuss my experience with the senolytics dasatinib, quercetin, and roxithromycin in detail here: Eliminating Zombie Cells – My Protocol for Fighting Senescent Cells
MAO-B inhibition
The catecholaminergic neurons (dopaminergic & noradrenergic neurons) are among the most rapidly dying neurons in the human brain. In fact, it is thought that we lose about 10% of the originally half a million or so dopaminergic neurons per decade.
The ongoing loss of catecholaminergic activity not just progressively diminishes drive, energy, and zest for life, but also sets a limit to the upper human lifespan as these neurons are required to keep many parts of the nervous system functioning.
If more than 80% of these neurons are dead, it is called Parkinson’s disease, which is often prefigured by depression. Inhibition of MAO-B is known to drastically slow the loss of dopaminergic neurons, as DA neuron death is in part a consequence of reactive molecules produced by MAO-B. In the past, I was taking low doses (0.05mg per day) of rasagiline. Unfortunately, the increase in dopamine levels turned me into an asshole, so I reduced my dosage to 0.02mg per day (I make microdoses myself using a morter, methylcellulose, and a capsule machine).
Optimizing sleep
Obviously, sleep is crucial to many aspects of brain health, ranging from housekeeping functions that cannot be carried out during wakefulness, to glymphatic outflow of waste products, to neuroplasticity.
I usually go to bed at the same time, make sure my room is dark and quiet, take a couple of supplements (magnesium, taurine, glycine), and expose myself to blue light in the morning.
In the past, I have taken sleeping pills for some time, which though was presumably a bad idea because a variety of CNS depressants (anticholinergics, antihistamines, GABAergics) are associated with dementia progression in ways that seem not accounted for by residual confounding.
I discuss my sleep optimization routine in more detail here. I discuss hypnotics in more detail here.
“Brain stimulation”
Keeping the brain active and engaged is probably the most important of all. I keep my brain active by exercising, continuously learning and doing new things, having meaningful social relationships, and creating purpose for myself. I wish I could travel more (“environmental enrichment”). The anti-neurodegenerative effects of these “interventions” are just a nice add-on.
Whether psychedelic drugs are more hype than hope, or worse, whether they do more harm than good remains to be seen.
I discuss these and other matters in more detail here: Intelligence Enhancement
Sources & further information:
- Podcast: Peter Attia & Richard Isaacson: Alzheimer’s prevention
- Scientific article: Rapamycin and Alzheimer’s disease: Time for a clinical trial?
- Scientific article: Major Neurocognitive Disorder (Dementia)
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part IV: My Approach to Hormone Optimization for Longevity
In my biochemistry classes, I learned a lot about cellular biology. For example, I learned about genetics, and I learned about a large variety of intracellular processes orchestrated by weird-sounding proteins.
Unfortunately, for the most part, my professors failed to mention that we are not solitary single-celled organisms but rather a collection of billions of intricately interdependent cells that cooperate for a common goal (i.e., to pass on my genes effectively to the next generation).
To ensure proper cooperation of these individual cells, early animals evolved a large number of signaling molecules to instruct each other on how to behave under certain conditions in order to serve the greater good (i.e., the survival and reproduction of the organism).
If these molecules are used for sending signals between cells over long distances (e.g., in vertebrates via the bloodstream) these molecules are called “hormones”, which derives from ancient Greek and means “to set in motion”.
For about 500 Mio. years, hormones have been coordinating the behavior of individual cells throughout the animal kingdom. In vertebrates, there are a vast number of different hormones, each for a distinct purpose. Some of these hormones act at the most powerful layer of intervention – namely, to alter the expression of genes directly.
Hormones are not everything, but without proper hormonal health, everything is nothing. Unfortunately, hormone replacement is a very neglected area in the conventional healthcare system.
I discuss some of the most important hormones, and my experience with them, here.
Hormones & aging
As I age, hormone levels decline across the board and my cells are progressively deprived of these crucial intercellular mediators.
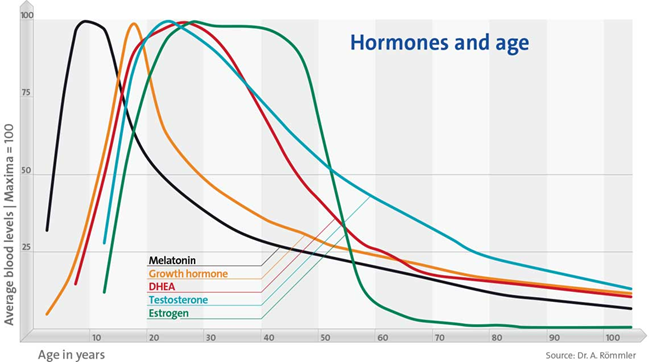
While the age-related hormonal decline is “natural”, evolution never prepared humans to be much older than age of 40. The hormonal decline is simply a byproduct of the aging process, including a deterioration of the hypothalamus, pituitary gland, peripheral glands, and target cells. In my opinion, restoring hormones to youthful levels is just as unnatural as living beyond the age of 50 anyway.
Proper hormone levels are crucial for graceful aging. Hormones are needed to help with nutrient partitioning, energy generation, musculoskeletal health, brain function, heart health, and pretty much everything my body does. Furthermore, hormones are intricately linked to vitality.
To me, longevity means much more than simply maximizing my lifespan. Above all else, it means maximizing my quality of life. If I had to choose, I would choose vitality over lifespan any day. In my opinion, achieving excellent health, performance, and well-being beyond the age of 50 is hardly possible without the use of at least some exogenous hormonal support – at least for most people.
- First, hormones are important for maintaining the structure and function of all of the tissues and organs in my body.
- Second, hormones have a major say in how I feel, which then co-determines the lifestyle choices I make.
Hormones & lifespan
I will now briefly discuss various hormones and how they relate to different aspects of the aging process. This list is far from exhaustive.
Cortisol
An aquiantance of mine who used high doses of hydrocortisone (25mg/d) noticed his hair greying. After stopping the hydrocortisone, the greying reversed again.
Cortisol is a steroid hormone that co-regulates about 20% of the genome. The primary purpose of cortisol is to prepare the mind and body for action and stress.
The amount of cortisol someone produces is one of the many variables determining how fast someone ages. At high levels, cortisol has pro-aging effects on every cell of the body because cortisol downregulates the expression of a number of genes involved in cellular repair, anabolism, and autophagy.
There are two closely related species of opossums with almost the exact same genetic makeup. However, one species has a 20% longer lifespan than the other (under laboratory conditions). It turns out, that the faster-aging species evolved to have substantially higher levels of cortisol because it lives in a much more dangerous habitat (predation) and it is thought that the higher levels of cortisol partially explain the faster rate of aging.
Higher levels of cortisol may also be part of the reason why some females seem to have aged five or more years over the course of a single pregnancy, a time during which cortisol levels are double to quadruple their normal levels. Speculatively, cortisol may also be part of the reason why the body of 39-year-old MLKJ reportedly looked like the body of a 70-year-old during autopsy. Due to the intense pressure he was under, his adrenals have probably been secreting vast amounts of cortisol for many years.
There is a reason for the saying that “stress can kill you”. Well, it is not the “stress”, but rather the associated hypercortisolemia.
Thyroid hormones
Thyroid hormone’s primary role is to increase the metabolic rate of every cell in the body. In simple terms, thyroid hormones make everything faster. For example, this includes the rate at which nutrients are metabolized, neurons communicate, hairs grow, or wounds heal. All else being equal, thyroid hormones also increase the speed of aging, and there are correlations between having lower levels of thyroid hormones and being a centenarian.
However, things are more complex than this. Thyroid hormones are highly anti-atherogenic. Furthermore, thyroid hormones stimulate the immune system and also reduce the risk of neurodegeneration. Moreover, higher thyroid hormones are correlated with greater vitality, which then affects lifestyle choices, which then affect the aging process.
In sum, while higher thyroid hormones may decrease maximum lifespan, higher thyroid hormones may have a protective effect against some usual causes of death, and thyroid hormones may also increase health span.
Growth hormone & IGF-1
Growth hormone & IGF-1 are in some respects similar to thyroid hormones in terms of lifespan & health span. By stimulating the ras-PI3K-mTOR pathway, GH & IGF-1 may decrease maximum lifespan.
Daf2 gene knockout (IGF-1 receptor) in C. elegans increases lifespan by almost 100%. Increased activity of the ras-PI3K-mTOR pathway may also be the major reason why larger dogs have shorter lifespans than smaller dogs. Furthermore, higher levels of GH & IGF-1 or insulin are associated with a greater incidence of cancer.
However, higher levels of IGF-1 are also associated with better metabolic health, less atherosclerosis, cardioprotection, better kidney health, better joint and bone health, greater muscle mass, and less dementia. Furthermore, GH and IGF-1 have potent effects on energy levels, mood, well-being, cognition, vigor, and vitality. All of these affect the lifestyle choices we make…and around 80% of us will be killed by diseases that are tightly linked to lifestyle.
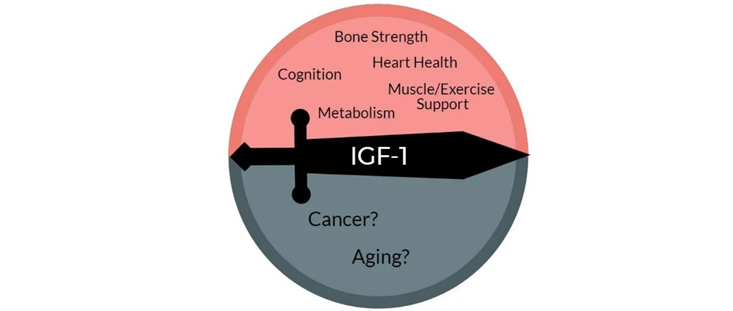
When it comes to the “ideal” IGF-1 levels, good things seem to happen at both ends of the U-shaped curve. An IGF-1 towards the lower end disinhibits autophagy and stem cell proliferation. An IGF-1 towards the higher end stimulates tissue maintenance, repair, and growth. Thus, instead of aiming for IGF-1 levels that are constantly low (like while fasting) or constantly high (like when taking GH), the best approach is presumably to cycle IGF-1 levels from low to high.
Tactics: My current approach to hormone replacement
Tweaking my hormones has taught me how much my mood & functioning are co-determined by my hormone levels at any given point in time. I discuss some of my experiences with hormones in more detail here.
Currently, I supplement with a low dose of metreleptin and I am on my TRT Lite protocol.
My IGF-1 is still fairly high and I will consider using 1IU of growth hormone in a couple of years from now.
Strategies I follow to optimize my hormones naturally
- I optimize my sleep.
- I make sure I eat enough calories (about 3500-4000kcal per day), with enough good carbs and good fats. Whenever my caloric intake is insufficient, some of my hormones decline, including sex hormones, thyroid hormones, and IGF-1.
- I stopped any kind of fasting (both multi-day fasting as well as intermittent fasting) in part because fasting can cause all sorts of hormonal issues.
- I put a prime on exercise – but I make sure not to overdo it.
- I used to make sure that I do not dip below 14% body fat. Whenever I did, a number of hormones took a hit. Starting semaglutide allowed me to tolerate about 2% lower levels of body fat without hormonal deterioration but I am currently off it. I now circumvent this problem altogether by taking a low dose of metreleptin sufficient to mimic a body fat percentage of about 15-20% (but without the aesthetic and metabolic downsides). Furthermore, leptin has many great metabolic effects, particularly if insulin sensitivity is high at the same time.
- I take a couple of supplements to prevent nutrient deficiencies (discussed here).
- I run an extensive hormone panel once per quarter (discuss here).
I discuss my personal experience with hormones in more detail here.
Sources & further information
- Scientific article: The physiology of endocrine systems with ageing
- Podcast: Peter Attia – The endocrine system: exploring thyroid, adrenal, and sex hormones
- Opinion article: Dr. Linder – Hormones
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part V: Crucially Important & Underrated – My Protocol for Optimizing Metabolic Health
Metabolic health is intricately connected to everything. For this article, I want to give credit to Dr. Peter Attia. I learned more from his podcast The Drive than from my biochemistry classes.
What is metabolic health?
It is hard to define what metabolic health is. It is easier to define it by what it is not. Metabolic health is the opposite of metabolic syndrome, which is essentially a disruption in proper energy metabolism. In practice, metabolic health is somewhat synonymous with “being highly insulin sensitive”, which has to do with a myriad of different factors ranging from muscle mass, exercise status, inflammation, and a plethora of genetic and hormonal factors, some of which are discussed in more detail shortly.
Metabolic health is a continuum. At one end, there are highly metabolically healthy individuals with great insulin sensitivity, metabolic flexibility, and nutrient partitioning. On the other end, there is full-blown type 2 diabetes.
In the industrialized world, currently, around 10% of the adult population is suffering from type 2 diabetes (but only about half are aware of it). Due to an aging population, more sedentary lifestyles, and increasing adiposity, the prevalence of this disease is expected to double within the next twenty years or so. Many more people are currently prediabetic and/or have “metabolic syndrome”.
Someone is “diagnosed” with metabolic syndrome if they have at least three of the following:
- hyper-glycemia
- hyper-tension
- hyper-lipidemia
- hyper-weight
Down the line, most people with metabolic syndrome will end up with serious conditions such as heart disease (including heart attack and stroke), type 2 diabetes, chronic kidney disease, cancer, or dementia.
Together, these constitute the most common causes of death worldwide. Metabolic syndrome was also one of the most important underlying factors that caused people to succumb to COVID-19, which illustrates the importance of metabolic health for health and longevity.
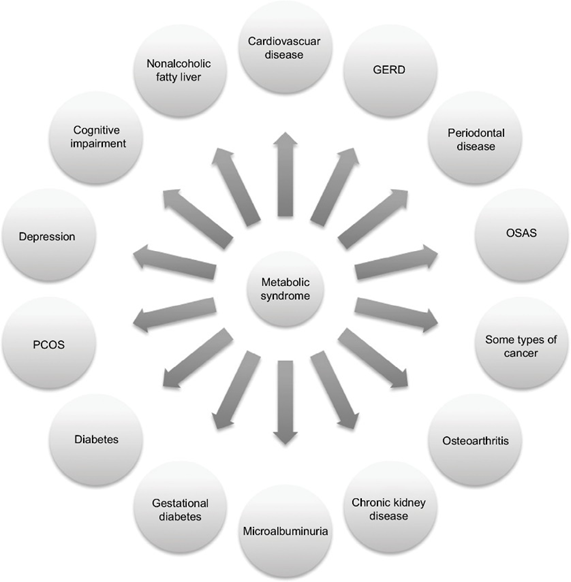
Metabolic health depends on a number of things. These include genetics, exercise status, a variety of different hormones, muscle mass, and body fat levels (particularly the amount of visceral adiposity). These factors can tilt metabolism towards a more healthy or a less healthy state.
Tactics I follow to promote metabolic health
- Muscle mass
- Endurance exercise
- A note on VO2 max
- Hormone optimization
- Metabolic drugs
- My experience with fasting
- (Mostly) avoiding sugar
- Thoughts on carbohydrate intake
- (Mostly) avoiding dairy
- Occasionally wearing a CGM
- Sleep optimization
- Thoughts on omega-3 & omega-6 fatty acids
- Keeping body fat levels low
- Leptin
- Supplements
- Are drugs necessary?
Muscle mass
I try to maintain a decent amount of muscle mass. To do so, go to the gym a couple of times per week. Muscle mass is fundamentally acquired by progressive overload and a slight caloric surplus. Obviously, I “never skip leg day” because legs have by far the biggest muscle groups in the body (at least in humans). My TRT Lite protocol surely helps.
Having a decent amount of muscle mass is known to improve every aspect of metabolic health. Muscle is a very insulin-sensitive tissue that will function as a “glucose sink”, therefore improving insulin sensitivity and nutrient partitioning. The more muscle, the more glucose and fatty acids are mopped up by muscle tissue at rest, particularly during the hours post-workout.
Therefore, all else being equal, more muscle equals greater insulin sensitivity.
I discuss the effects of exercise on vitality (energy levels & mood) in more detail here.
Endurance exercise
Zone II exercise is cardiovascular exercise performed right at the cusp before lactate starts to accumulate. Said another way, it is exercise during which oxidation of fatty acids is at its maximum. Through a variety of signaling pathways, zone II exercise stimulates mitochondrial function and mitochondrial biogenesis, which vastly improves metabolic health through a variety of mechanisms.
A couple of times per week (depending on the season) I do a dedicated session of zone II cardio. Below is a screenshot from my Polar data of what a zone II session looks like for me (I usually end with a 5min high-intensity part). My average heart rate is between 70-80% of my maximum heart rate (which is a little over 180bpm).

A proper endurance exercise session is known to improve insulin sensitivity for at least 24 hours. I am always amazed at how potent this is when I look at my CGM data. For example, eating pizza after a heavy endurance session has much less of an effect on my blood sugar compared to eating pizza without having exercised that day.
Exercise, in particular endurance exercise, leads to countless adaptations in skeletal muscle. One of the presumably most important changes is an improvement in mitochondrial function and stimulation of mitochondrial biogenesis. Said another way, endurance exercise increases both the number as well as the quality of mitochondria, which have a variety of positive effects on metabolic health (and a number of other things).
Furthermore, endurance exercise stimulates an insulin-independent GLUT-4 translocation, which will reduce blood glucose levels in an insulin-independent way.
Moreover, endurance exercise leads to a depletion of intracellular nutrient stores (particularly glycogen), which activates a metabolic master switch enzyme called AMPK with downstream effects of increasing insulin sensitivity.
On an epigenetic level, endurance exercise also leads to altered expression of a large number of genes.
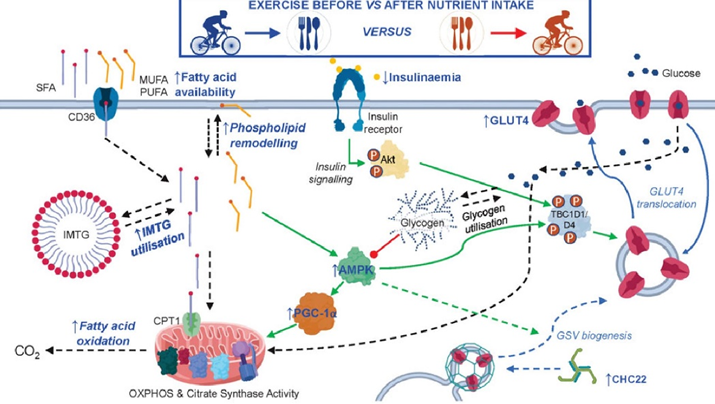
Because of these changes, a single endurance exercise session increases insulin sensitivity for many hours to come.
Furthermore, by regularly depleting glycogen stores (e.g., by regularly doing endurance exercise), dietary glucose is handled the way it should be, namely, by being funneled into hepatic or muscle glycogen synthesis. Conversely, if glycogen stores are always tapped out, excess glucose is biochemically much harder to manage and comes with a host of unfavorable biochemical alterations.
In sum, the end result is that endurance exercise has an incredible effect on insulin sensitivity and mitochondrial function, both of which are at the center of metabolic health.
A note on VO2 max
VO2 is a measure of how much oxygen (and therefore metabolic substrates) my muscles are capable of using during physical exertion, which is an indicator of how well mitochondria are performing. VO2 max is highly correlated with lifespan and health span, as “survival of the fittest” would predict.
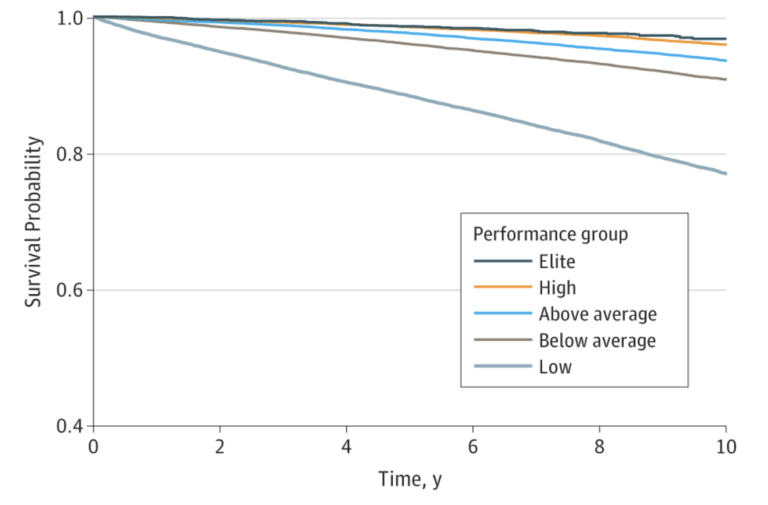
According to Peter Attia, the “ideal” VO2 max training is spending 80% of the time allocated to aerobic exercise in the zone II range (about 75-80% of maximum heart rate) and spending 20% in zone V (e.g., via HIIT training). According to Dr. Attia, the minimum effective dose is about three hours per week.
I end most of my cardio sessions with a 5-minute high-intensity part. I do not do more because I hate HIIT.
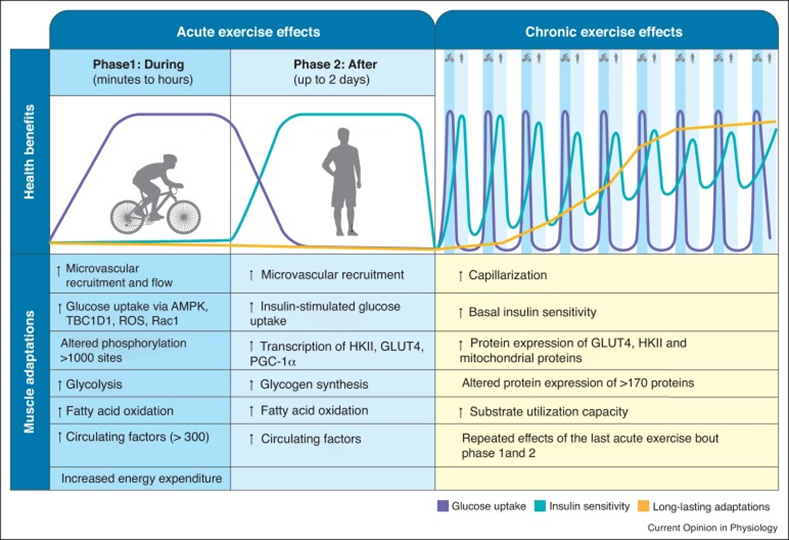
Hormone optimization
I watch a number of hormones closely, including sex hormones, thyroid hormones, and IGF-1. Having “youthful” levels of these hormones will improve nutrient partitioning, insulin sensitivity, muscle growth, keep body fat reasonably low, and hundreds of other things.
Furthermore, hormones have powerful effects on energy levels, mood, vigor, and vitality, and therefore, they make leading a healthy lifestyle much easier.
Currently, I only supplement with HCG and a low dose of metreleptin.
Metabolic drugs
For multiple years, I took a low dose of semaglutide. I mostly used semaglutide to reduce my appetite, which I find distracting. Semaglutide has significantly increased my quality of life. I discuss my experience with it in more detail here. For the past 2 years I have been off it because it lowers my appetite too much, even at very low doses. weight gaining impossible.
For most people, semaglutide increases metabolic health because it leads to a loss of body fat, which increases insulin sensitivity. Some people worry that it decreases muscle mass. Personally, I have built about 5 kg of muscle on semaglutide, though I also started doing CrossFit at about the same time.
Because my levels of uric acid are generally on the higher end, I also take allopurinol (150mg/d) to reduce my uric acid to the 4-5 mg/dl range.
In the past, I have also experimented with acarbose, metformin, and SGLT-2 inhibitors, but for me, all three had more downsides than upsides. I am currently also off rapamycin, which undoubtedly affects (improves?) metabolic health.
I discuss a variety of metabolic drugs, and my experience with them, in more detail here.
Fasting
Intermittent fasting and multi-day fasting are both known to improve metabolic health through a variety of mechanisms. For example, both are known to reduce mTOR activity and upregulate AMPK activity, improve insulin sensitivity, stimulate autophagy, and promote metabolic flexibility.
As always, many processes happen in parallel and a plethora of changes are cross-interacting, influencing, and driving each other. Parallelism is something our brains are just awfully bad at grasping because we evolved to cognize linearly in space and time.
However, for me, all kinds of fasting did more harm than good. I discuss my experience with intermittent fasting here, and my experience with multi-day fasting here.
Mostly avoiding sugar
A couple of years ago, I was low-level addicted to sugar, in particular chocolate. Interestingly, since going on semaglutide it seems that sugar, and sweetness in general, has lost most of its “pleasure” (though, I can still taste it normally – just without much “pleasure”). Interestingly, this effect persisted even after coming off semaglutide.
Sugar in the way it is currently consumed, is addictive and causes weight gain and creates all sorts of metabolic issues. Fructose metabolism significantly differs from glucose metabolism, often leading to intrahepatic fat accumulation, which then drives insulin resistance, metabolic disease, and obesity.
Obesity comes down to two things:
- 1) People eat too much (e.g., due to boredom, psychological eating, constant availability of highly palatable foods rich in fat & sugar & salt).
- 2) People store too much (regardless of how much they eat).
By hijacking the reward system (“people eat too much”) and causing insulin resistance (“people store too much”), sugar hits both.
Interestingly, on a diet high in fructose, many people get insulin resistant and develop a fatty liver (NAFLD) even if they do not gain weight. Likewise, many people have a dramatic improvement in metabolic health & NAFLD if they simply switch out fructose for glucose. For this very reason, some researchers call fructose “ethanol without the buzz”, as it leads to non-alcoholic-fatty-liver disease (NAFLD) in a similar way alcohol leads to alcoholic fatty liver disease (AFLD).
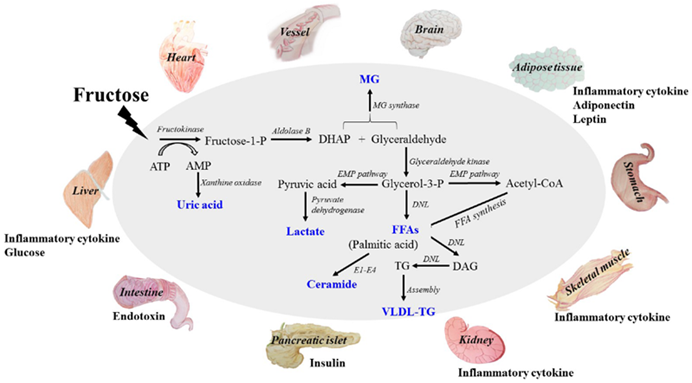
Furthermore, fructose is thought to elevate levels uric acid levels, which not only elevates blood pressure but also seems to crystallize in the form of calcium urate crystals, gradually over decades leading to low-level organ damage (much more than just gout).
What about fruit? Consuming fruits in reasonable quantities is probably fine as the fructose contained is not very concentrated. Furthermore, there is an adequate amount of fiber contained within most fruits, which slows stomach emptying and enzymatic breakdown. As a result, lots of the fructose in fruit is metabolized to glucose in the intestinal tract long before it can overrun the liver.
Blockers of fructose metabolism are currently in clinical development by Pfizer.
I discuss sugar in more detail here.
Avoiding alcohol
In terms of metabolic health/disease, ethanol is quite similar to fructose. It worsens metabolic health and leads to intrahepatic lipid accumulation, which causes insulin resistance.
Furthermore, alcohol worsens sleep, which is also important for metabolic health because firstly, the nervous system regulates cortisol secretion (which has dramatic effects on metabolism) and secondly, the nervous system regulates body weight and insulin secretion.
I find that already the slightest amount of alcohol destroys my sleep parameters (SWS, REM, RHR, HRV, body temperature patterns), at least as measured by my Oura ring. I used to party a lot when I was younger but at this point in life, “sacrificing” the next day is not worth it anymore. I only drink it on very special occasions but then I really enjoy it.
Carbohydrate intake
I feel and function best if I titrate my carbohydrate intake to my leanness and activity levels. So, usually, I eat quite a lot of carbs (about 300-400g per day) because I simply feel and function better if I eat them. I find that if I do lots of strenuous activity (e.g., CrossFit) I can tolerate a lot more carbs because they are immediately burned (or absorbed) by muscles.
Similarly, the leaner I am, the more carbs I need to not experience a decrease in energy levels. This is likely because insulin is “the hormone of abundance” and signals to various brain nuclei (first and foremost hypothalamic POMC neurons) that there is no reason to worry about starvation.
I believe that low-carb diets are great for people who are overweight and/or metabolically deranged, but they may do more harm than good for people who are already lean and insulin-sensitive.
I discuss my approach to macronutrients in more detail here.
“Bad” carbs
In the past, I tried to “hack” this by using acarbose or SGLT-2 inhibitors, both of which reduce post-meal glucose excursions. However, acarbose consistently spiked my liver enzymes and SGLT-2 inhibitors consistently dehydrated me. Needless to say, I stopped taking both. I do not worry too much about “bad carbs” but I mostly avoid overly sugary carbs.
Dairy
For many people, dairy may be inflammatory, drive gut issues, and seems to negatively affect the lipid profile and metabolic health. This might be due to some of the protein fractions in diary (i.e., whey & casein) being hormonally active (i.e., possibly, they are transcytosed and act on hepatocytes).
I personally eat a lot of dairy, sometimes exceeding 2l of whole milk or kefir a day, which is my favorite drink of all time.
I discuss dairy in more detail here.
CGM
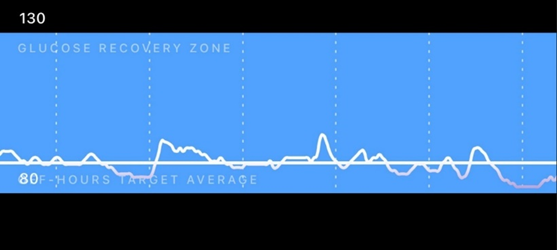
From time to time, I carry a continuous blood glucose monitor (CGM) to see whether my diet needs some changes. Below is a screenshot of a day that went particularly well.
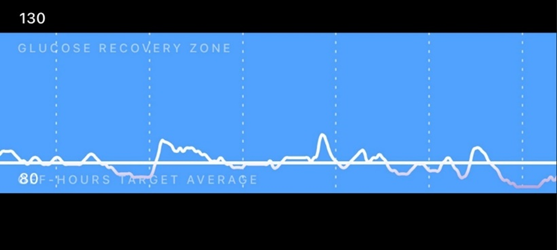
Through CGM use, I found out that I am very insulin sensitive, and on self-performed OGTTs I am back to baseline after about 20-40 minutes (usually undershooting my baseline slightly). I attribute this to my leanness and high activity levels.
Sleep optimization
There are multiple ways in which bad sleep hampers metabolic health.
- In a sleep-deprived state, cortisol levels are generally high. Cortisol has a huge effect on various aspects of metabolism. In Cushing syndrome (excess cortisol levels) metabolic health is usually crap.
- Good sleep is necessary for proper growth hormone release. Growth hormone is anabolic to muscle and catabolic to fat, particularly visceral fat (discussed shortly).
- The nervous system itself is quite a powerful regulator of metabolic status. For example, through vagal nerve endings, the nervous system affects liver metabolism and insulin secretion. Bad sleep is usually associated with greater basal insulin secretion, likely independent of cortisol and presumably mediated by vagal control of the pancreas.
- In a sleep-deprived state, willpower and metacognition are poor, which leads to bad food choices and forgoing exercise, both of which are causally related to metabolic health.

I discuss how I optimize my sleep in more detail here.
Omega fatty acids
Omega-3: I make sure I get enough Omega-3 fatty acids. I supplement with 2g of EPA/DHA per day. I describe them here.
Omega-6: While the science is inconclusive, it seems that high amounts of omega-6 fatty acids may promote inflammation by being converted into pro-inflammatory prostaglandins. Therefore, some researchers believe that it is best to avoid excessive consumption of vegetable oils (e.g., soy, corn, sunflower) because of their high omega-6 fatty acid content. I find this to be a huge nuisance in modern life, and I pay little attention to it. I wish food manufacturers would just switch out sunflower oil for canola oil, which would (likely) make our lives a little healthier.
Keeping body fat levels low
One of the most powerful interventions to improve metabolic health is to keep body fat levels low. I personally keep my body fat in the 10-12% range and measure my body fat once in a blue moon via a DEXA scan. I discuss body fat in more detail here.
Tools I use for keeping body fat low include exercise and a very low dose of metreleptin. The addition of a low dose of metreleptin allows me to keep a low body-fat percentage without suffering from starvation-related adaptations.
The less adipose tissue there is, the more “primed” adipose cells are to store fat, and the lower the adipose tissue-mediated inflammation. This means, that all else being equal, a lower body fat percentage is associated with less inflammation, better insulin sensitivity, and improved nutrient partitioning. The vast majority of people with little adipose tissue have decent to great metabolic health (some Southeast Asian ethnicities may be an exception).

In particular, ectopic fat (fat stored in non-fat cells) and visceral fat (fat stored around the liver) is the metabolic devil, causing inflammation and insulin resistance. Both are highly correlated with increased levels of obesity. And since visceral fat is, for the most part, a function of body fat percentage, keeping body fat levels low is one of the most effective ways to prevent visceral fat accumulation.
Visceral adiposity leads to a fatty liver, which wreaks havoc on all aspects of metabolic health, particularly insulin sensitivity. The science is quite complex and has to do with local adipokine signaling, the local elevation of free fatty acids activating the inflammasome, and free fatty acids inducing insulin resistance. I discuss visceral fat, and potential ways to reduce it, in more detail here.
Leptin
Leptin is a very misunderstood hormone. It has little to do with being “the satiety hormone” as it is commonly believed. Rather, the purpose of leptin is to protect the animal from starvation. Because my body fat is lower than my body would like it to be (about 10-12%), my leptin levels are naturally very low (less than 1ng/dl).
Leptin has a huge effect on metabolism. Firstly, it is a master-regulatory hormone that affects hypothalamic and pituitary function on a broad level. In a state of hypoleptinemia, hypothalamic functions such as thermoregulation, sleep, sex drive, energy metabolism, appetite, sympathetic nervous system activity, and hormone secretion are all affected in a way to prime the organism to conserve energy and to store fat.
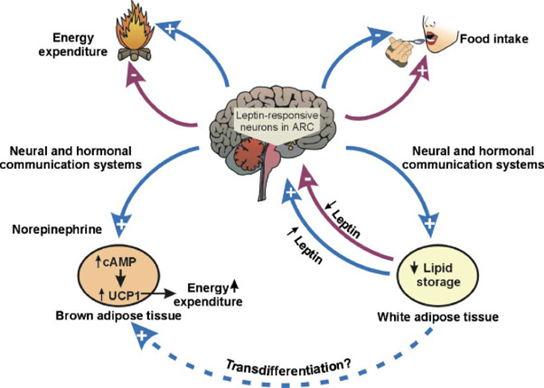
Secondly, leptin acts directly on the liver, adipose tissue, and muscle cells, among others, instructing them to alter intracellular metabolism. For this reason, leptin analogs or leptin receptor agonists are promising interventions to treat non-alcoholic fatty liver disease. Hypoleptinemia may also be the reason why some lean and healthy individuals sometimes have erratic blood sugar patterns and some other metabolic (and hormonal) issues.
Leptin expression and secretion are highly correlated with the amount of adipose tissue someone carries, particularly subcutaneous adipose tissue.
To increase my endogenous leptin secretion (without gaining weight), for a couple of months in the past I have topically applied a self-made cream containing the PPAR-gamma agonist pioglitazone (which induces preadipocyte differentiation) to my upper legs with the goal of increasing the number of local subcutaneous adipocytes to preferentially shift fat storage towards this region. I did this to create my own endogenous “leptin factory” in a place that is optically not displeasing.
Ever since my upper legs have now a slightly thicker layer of subcutaneous adipose tissue. As a side note, I suspect that pioglitazone cream could potentially become the next anti-aging cream because of its ability to counteract, and perhaps even reverse the age-associated loss of facial fat. Unfortunately, my leptin levels only rose barely or not at all.
I decided to hack my way around gaining fat by jumping on a very low dose of metreleptin sufficient to mimic a body fat percentage of 20% (but without the aesthetic and metabolic downsides of actually being 20% body fat). Interestingly, from that point on my blood glucose became much more stable (among many other things).
The mental changes were profound. I plan on writing an article about this in the future.

Using metreleptin allows me to carry a lower-than-ideal body fat percentage without any soul-sucking starvation-related adaptations (e.g., mood, energy, sex drive, sleep, hormonal health) while benefiting from the aesthetic and metabolic-health-improving effects of having low body fat.
It is no rocket science to infer that leptin agonists could potentially revolutionize bodybuilding and a fitness industry focused on being “shredded”.
I discuss leptin, and my experience with it, in more detail here.
Supplements
I am quite sure that compared to the other tactics on this list, supplements do comparatively quite little. Most of the supplements I take are taken with a “just-in-case” mentality, and I am equally uncomfortable taking them compared to not taking them. Nonetheless, I have been taking a variety of supplements for about 4-5 years now, and nothing “bad” has happened.
I discuss the supplements I take, and why I take them, in more detail here.
Are drugs necessary?
It might be obvious by now that I have a fondness for pharmacology. However, I am pretty sure that I (and presumably most other young healthy people who have not been overweight during childhood or adolescence) could achieve pretty decent metabolic health by lifestyle changes alone, including a hefty dose of exercise, keeping body fat levels fairly low, eating healthy, avoiding sugar, avoiding dairy, and optimizing sleep.
However, some of the same signaling cascades that are activated by lifestyle choices can also be manipulated pharmacologically. In principle, my cells could not care less whether downstream signaling pathways are activated by lifestyle choices or pharmaceutical intervention.
I discuss my thoughts on the “ethics” of intervention in more detail here.
For a variety of reasons, I choose to take metabolic drugs even though they are not necessarily necessary. All five drugs I regularly take are, to some extent, metabolic drugs. These include thyroid hormones, semaglutide, metreleptin, allopurinol, and rapamycin. However, these drugs are by no means a replacement for the lifestyle changes discussed above.
Other articles
Because metabolic health is a huge (and hugely important) topic but this article is already awfully long, I wrote three articles on the subject that will go in much more depth:
For more info on muscle mass & longevity, I highly recommend Outlive by Peter Attia.
Sources & further information
- Podcast: Peter Attia & Richard Miller: The gold standard for testing longevity drugs: the Interventions Testing Program
- Podcast:Peter Attia & Ronesh Sinha – Insights into the manifestation of metabolic disease
- Scientific study:The acute vs. chronic effect of exercise on insulin sensitivity: nothing lasts forever
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part VI: What Is All the Fuss About mTOR?! – Why (and How) I Reduce the Activity of the mTOR Pathway
In eucaryotic organisms (plants, animals, fungi, protists), the mTOR pathway is a central regulator of cellular metabolism, growth, and survival. It has long been postulated that mTOR activity is one of the central control knobs of the speed of aging, which can be regarded as a continuation of the pre-adulthood growth program.
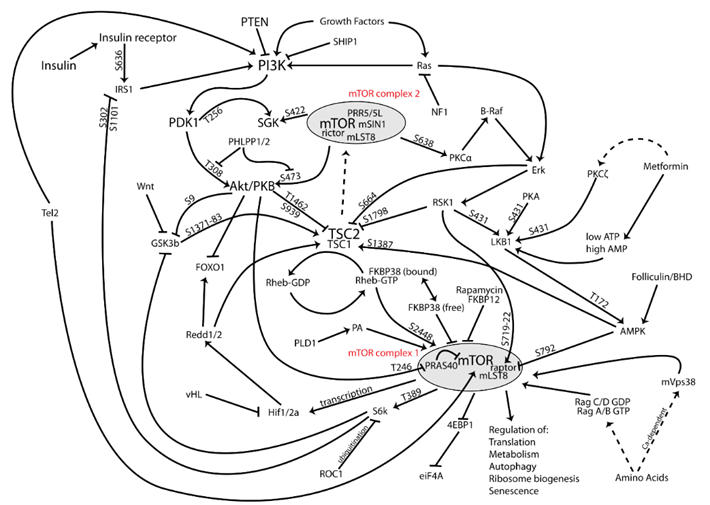
When there are sufficient nutrients & amino acids (glucose, leucine, arginine, methionine) and sufficient extracellular growth signals (e.g., insulin, IGF-1, tissue-specific growth factors), the mTOR complex is activated and drives a host of anabolic processes, including growth and development.
However, because animals usually die long before aging kills them, evolution never bothered with reducing the activity of the mTOR pathway after sexual maturity had been reached. Thus, to the detriment of the old animal, mTOR is set at too high a level after growth and development have been completed.
mTOR inhibitors are a promising pharmacological fix for the problem evolution never bothered to confront.
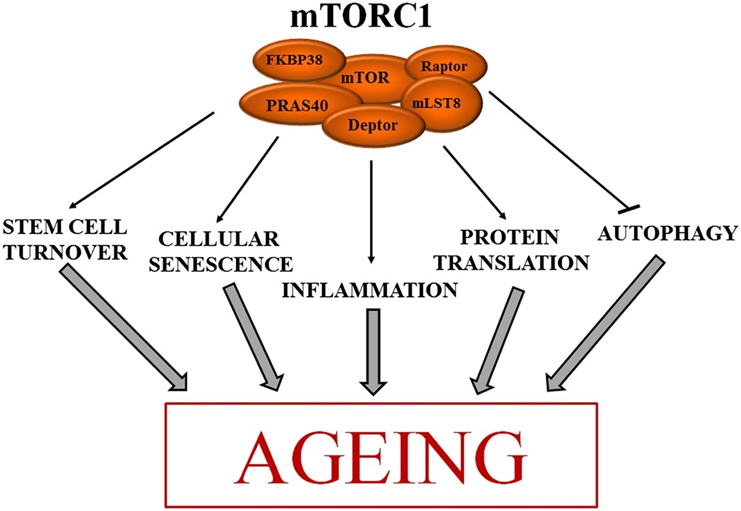
Tactics to keep mTOR activity (fairly) low
- My thoughts on fasting & caloric restriction
- Metformin
- Rapamycin
- IGF-1 – A double-edged sword
Fasting & caloric restriction
Fasting and caloric restriction are powerful anti-aging interventions in almost every animal. It is thought that both exert many of their beneficial effects partly through mTOR downregulation. However, for me, both fasting and caloric restriction were a dead end.
Fasting and CR are not just unpleasant, but they might drive peripheral thyroid resistance, alter brain energy homeostasis, and cause a sustained reduction in IGF-1 and leptin signaling. Furthermore, both reduce vitality. I want to enjoy my life, and feeling cold, lethargic, and hungry all the time is not an option.
I discuss my experience with multi-day fasting in more detail here.
I discuss my experience with caloric restriction in more detail here.
Metformin
There is evidence that metformin indirectly inhibits the mTOR1 complex. In the past, I had been on metformin for about two years but I stopped because of a number of reasons.
I discuss my experience with metformin in more detail here.
Rapamycin
I take 4 mg of the mTOR inhibitor rapamycin once per week, nine months on and three months off.
mTOR is a protein kinase that has a finger in every major process in the cell and is partly responsible for regulating “growth mode” vs. “conserve resources mode”. The promise of rapamycin is that it can give me most of the benefits of fasting without the downsides.
Rapamycin is an mTOR inhibitor, mimicking starvation pharmacologically including an artificial induction of autophagy. The cells in my body are tiny machines based on the laws of chemistry and physics and programmed by their genetic code. In principle, they could not care less whether effects come from endogenous molecules, exogenous molecules, or lifestyle changes.
Furthermore, and counterintuitively, I suspect that taking rapamycin is somewhat safer than doing regular fasts (discussed in more detail here).
Some people claim that it is better to take rapamycin less frequently (e.g., every other week but double the dose) to have proportionately more of it make it across the blood-brain barrier. For now, I just copy what most people at the frontlines do, namely taking rapamycin once weekly at a dosage of 4-8mg/week.
I discuss my experience with rapamycin in more detail here.
IGF-1 – a double-edged sword
My IGF-1 levels are slightly above the upper end of the clinical reference range naturally. IGF-1 is a protein quite similar to insulin and the two are phylogenetically related. Hence also the name: insulin-like growth factor. Furthermore, their receptors are very similar and IGF-1 even has a glucose-lowering effect mechanistically identical to insulin.
Both together are perhaps the two major systemic growth factors in the human body exerting their effect via powerful receptor-tyrosine-kinase-signaling, which downstream also activates the mTOR pathway. I discuss insulin signaling in more detail here.
IGF-1 is a double-edged sword. On the one hand, higher levels are associated with cancer (as it drives the mTOR pathway), and on the other hand, higher levels are associated with better mental health, cognition, vitality, muscle mass, bone structure, and a lower risk for dementia and atherosclerotic disease.
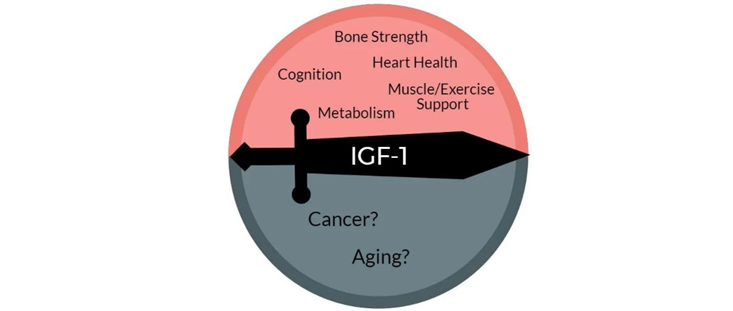
However, in part because my IGF-1 levels are naturally high, I try to keep AUC levels of insulin rather low by optimizing metabolic health (described here). Of note, insulin stimulates IGF-1 synthesis by the liver.
Sources & further information:
- Scientific article: Effect of rapamycin on aging and age-related diseases—past and future
- Scientific article: Cell senescence, rapamycin and hyperfunction theory of aging
- Scientific article: Rapamycin for longevity: opinion article
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part VII: Burning to Death – My Protocol for Lowering Inflammation
Over the last couple of years, more and more research has come out on how inflammation affects (and drives) a variety of conditions, ranging from dementia, osteoporosis, atherosclerosis, and depression.
Chronic inflammation has emerged as a key feature of aging and age-associated diseases (“inflammaging”).
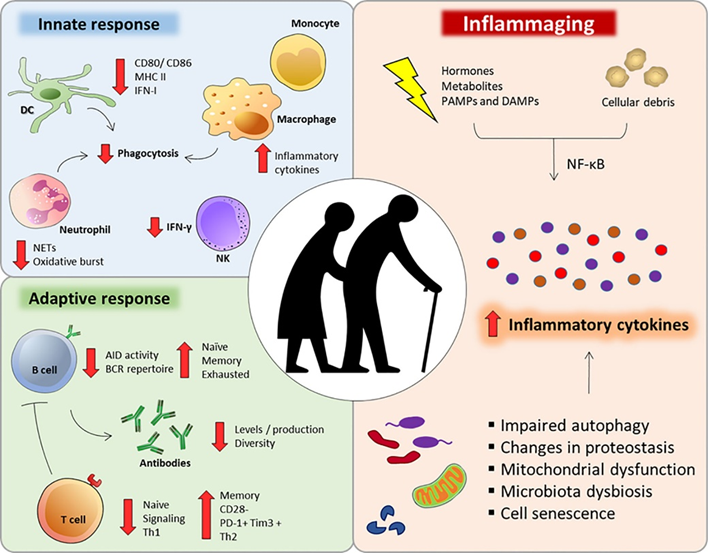
Age-related inflammation is thought to be caused by a variety of things:
- Senescent cells and their senescence-associated phenotype (SASP)
- Progressive accumulation of intracellular and extracellular waste products (e.g., advanced glycation end products, metals, particulate matter, microplastics, lipofuscin, etc.)
- Mutations to mitochondrial DNA (mitDNA) and mitochondrial breakdown
- Senescence of the adaptive immune system (immunosenescence) due to the depletion of hematopoietic stem cells and senescence of already existing lymphocytes, which causes the adaptive immune system to be hypofunctional, resulting in a hyperfunction of the innate immune system
- The herpesviruses EBV and CMV constantly trying to break out, which is followed by immune system activation. Beyond 60 years of age, roughly half of the human immune system is constantly busy with fighting CMV.
Because of these and other factors, background levels of inflammation keep rising as age goes up. Inflammation is then thought to harm tissues in at least three ways:
- Cytokines (inflammatory mediators) affect the function of healthy cells in a negative way. Particularly IL-11 has recently come out as a key player and preliminary data suggests that IL-11 blockade results in a lifespan extension of roughly 25%.
- Cytokines and inflammation-associated growth factors stimulate fibroblasts to build connective tissue. This build-up of extra connective tissue (i.e., fibrosis) affects the structural integrity of tissues.
- Cytokines hamper metabolic health directly by changing the activity and metabolism of several tissues.
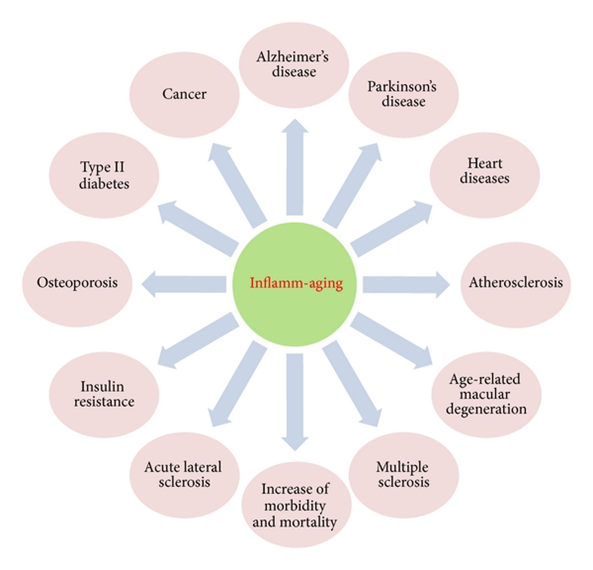
Tactics I follow to keep inflammation levels down
- Diet
- Daily exercise
- Optimizing metabolic health
- Keeping body fat low
- Rapamycin
- Counteracting the accumulation of senescent cells
- Antioxidants
- Niacin
- Beta-alanine
- Counteracting the accumulation of heavy metals
Diet
I follow a diet that is quite clean and rather low in pro-oxidative ingredients. During the day, I drink a lot of Huel shakes with olive oil. I discuss my diet in more detail here.
Daily exercise
I exercise daily. I discuss my exercise routine in more detail here: My Fitness Protocol
In the short term, exercise leads to a burst in oxidative stress due to the vast increase in metabolic rate. However, in the long term, exercise lowers inflammation (as measured in the form of lipid peroxidation and certain cytokines), presumably due to a counterregulatory upregulation of antioxidant systems and oxidative damage repair systems. It is thought that this effect is specifically pronounced with high-intensity exercise.
Hence, through defense mechanisms aimed at countering exercise-induced oxidative stress, cells and tissues in individuals who regularly exercise demonstrate reduced baseline oxidation levels. This concept is known as hormesis.
I personally end each zone II cardio session with 7 minutes of high-intensity exercise, which though is probably not sufficient for reaping the full benefits.
Optimizing metabolic health
I optimize metabolic health (see here).
Among other things, I try to keep the amount of visceral adipose tissue (a consequence of insulin resistance, alcohol, sugar, and cortisol) quite low as it drives inflammation and insulin resistance through the elevation of local free fatty acids and the secretion of proinflammatory adipokines and cytokines. I use a low dose of metreleptin to help with this.
Furthermore, I try to keep glucose excursions low because glucose spikes lead to the generation of advanced glycation end products (AGEs), which are then recognized by macrophages as a pro-inflammatory signal. I do this by eating/drinking a lot of Huel shakes and trying to keep high-starch meals to once per day (though with a lot of room for flexibility). On and off, I use a continuous blood glucose monitor for this.
I also experiment with a host of metabolic drugs. I currently take allopurinol. On and off, I also take microdoses of semaglutide. I discuss my experience with metabolic drugs in more detail here.
Lowering the activity of the mTOR pathway with rapamycin
mTOR activity stimulates inflammation, and inflammation stimulates mTOR activity. Rapamycin is pretty good at reducing age-related sterile inflammation. I take 4-5mg per week. From November to January each year I take a break, partially also because during this time people tend to get sick the most.
I discuss my experience with rapamycin in more detail here.
(By now it should be clear that many of the tools & tactics I use are beneficial for a variety of different strategies. For example, rapamycin keeps my cells from senescing, improves metabolic health (if dosed weekly), sharpens the adaptive immune system, reduces inflammation, improves cellular and tissue health, and counteracts carcinogenesis.)
Counteracting the accumulation of senescent cells
Senescent cells are proinflammatory because of the proinflammatory mediators they secrete. Rapamycin is particularly powerful in slowing the accumulation of senescent cells. Furthermore, I tried to clear some of my tissues of senescent cells by recurrent administration of dasatinib & quercetin (D&Q). I discuss my experience with senolytic drugs in more detail here.
Antioxidants
Ever since oxygen accumulated in the Earth’s waters and atmosphere about 2.5 billion years ago, the vast majority of complex life on Earth evolved to use oxygen for its metabolism. Unfortunately, the same oxygen is highly reactive and can damage all kinds of molecules. Thus, organisms evolved a variety of antioxidant defense mechanisms. When this balance is disrupted, it is called oxidative stress.
Reactive oxygen species (ROS) then damage cellular components, including lipids, proteins, and DNA. This then leads to dysfunction and inflammation. Furthermore, some of these destroyed components can hang around for years and decades (e.g., crosslinked collagen, lipofuscin, protein aggregates), impairing proper tissue function.
ROS specifically lead to mitochondrial damage because most ROS are generated in the mitochondria. This is one reason why the mutation rate of mitochondrial DNA is somewhere between a hundred and a thousandfold faster than nuclear mutation rates.
Antioxidants are molecules that either prevent reactive oxygen species from being formed or remove them (usually by directly reacting with them), thus minimizing their damage.
I take a couple of antioxidants in low doses. These include N-acetylcysteine (300mg), alpha lipoic acid (100mg), vitamin C (250mg), coenzyme Q10 (60mg), and selenium (100mcg). I discuss the supplements I have been taking now for roughly 5 years in more detail here.

Because ROS leak serves some physiological functions, including participation in intracellular signaling pathways, I am quite unsure about the benefits vs. harm of antioxidants. While antioxidants are perfect on paper, animal trials with antioxidants give conflicting results. I feel slightly less uncomfortable taking them vs. not taking them.
Another antioxidant (potentially) worth mentioning is resveratrol. However, in every experiment to date, resveratrol at several doses has failed to extend the lifespan of lean, genetically normal mice (with the exception of mice that were dying from a specific form of lipid poisoning due to being fed a high-fat diet).
Niacin
In the past I used to take niacin once daily (250mg – flush version). Niacin acts on the HC2A receptor (the same receptor ketone bodies act on) and has a variety of anti-inflammatory effects in a variety of tissues. However, niacin can cause insulin resistance for the next couple of hours, which is why I have stopped taking it.
Beta-alanine
I take beta-alanine (1g/d), which converts into carnosine. Carnosine not only protects against exercise-induced lactic acid production (as it buffers cells against changes in pH), but it also has metal-chelating effects, and some anti-glycation effects.
Glycation generates advanced-glycation end-products (AGEs), which are pro-inflammatory because they are recognized by macrophages as waste products (via the aptly titled “RAGE” – Receptor for Advanced Glycation End-products).

Counteracting the accumulation of heavy metals
Cadmium, lead, and mercury have no essential biochemical roles but exert a number of toxicities in multiple organ systems, in part because they create oxidative stress, which drives inflammation, and in part because they are mistaken by enzymes for essential cations such as calcium, magnesium, zinc, copper, and manganese.
I (try to) prevent the accumulation of heavy metals by taking a couple of supplements with metal-chelating effects (alpha lipoic acid, n-acetyl cysteine, beta-alanine, taurine). Some people claim that chelation therapy with EDTA or DMPS is much more effective. However, clinical data are lacking. Furthermore, some researchers speculate that EDTA and DMPS therapy frees heavy metals from bones, which are then inadvertently redistributed to the brain.
Bonus section: Inhibition of IL-11 signaling extends mammalian healthspan and lifespan
Lately, monoclonal antibodies targeting certain proteins have been all the rage. For example, antibodies targeting TNF-alpha have transformed the treatment of a variety of autoimmune diseases for the last 20 years or so. Or dupilumab (Dupixent), an antibody targeting interleukin 13 & 14, has transformed the treatment of atopic dermatitis and asthma.
Recently, an exciting paper was published. The authors blocked interleukin 11. Thus far, interleukin 11 has been elusive. It is thought that IL-11 mostly works in a paracrine fashion, meaning that it mostly acts locally on neighboring cells. When there is inflammation, among thousands of other things, IL-11 is secreted by cells to increase growth and inflammation in nearby cells, thus serving as an “inflammation amplifier” of sorts.
This makes sense in the context of injuries or infections. However, this is detrimental in the context of aging. Due to mitochondrial damage, accumulation of waste products, increased number of senescent cells, CMV and EBV trying to break out in a state of immunosenescence, etc. inflammation increases with aging.
In fact, aging is called “inflammaging” for a reason. The basic idea is that when one blocks IL-11, one decreases inflammatory processes, which are often associated with growth pathways (e.g., mTOR activation, AMPK downregulation, etc.). Therefore, by dampening the inflammation and growth through IL-11 blockade, this should theoretically increase health span and lifespan.
Lo and behold, in the paper published 10 days ago, lifespan and health span increased in a similar way as they do when rapamycin is administered (by roughly 25%). This finding, if confirmed by other laboratories (such as the Intervention Testing Program) is huge. I am eagerly awaiting results in genetically heterogenous mice, primates, and ultimately the approval for a human anti-IL-11-antibody (e.g., for fibrotic diseases).
Sources & further information:
- Scientific article: Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty
- Scientific article: Low-Grade Systemic Inflammation Connects Aging, Metabolic Syndrome and Cardiovascular Disease
- Scientific article: Chelation: Harnessing and Enhancing Heavy Metal Detoxification—A Review
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part VIII: My Physical Fitness Protocol
My grandpa recently died. He was in otherwise good health, but his hip joint was a mess due to years of hard work at his farm. Unfortunately, he was denied hip replacement. Consequently, he had a hard time walking and he started to just lie around in his living room (whereas before he was decently active). Not even one year later he died mostly because his cardiovascular system had deconditioned (“use it or lose it”) exacerbating congestive heart failure.
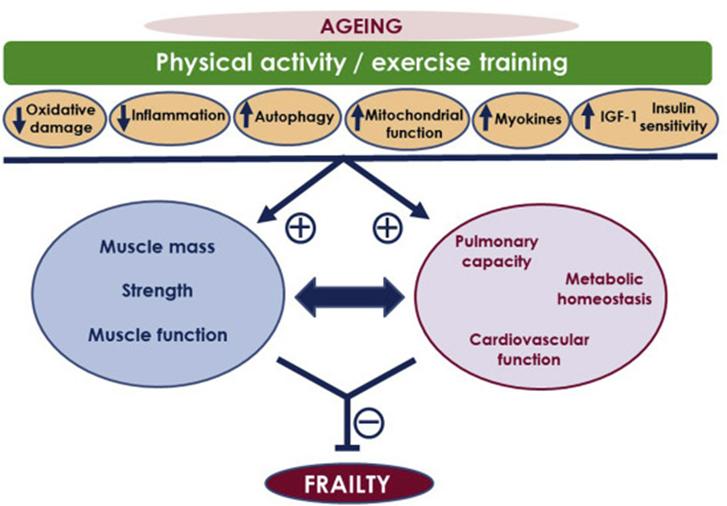
Similarly, even if metabolic health, neurobiochemistry, and what have you were “perfect” (whatever that means), if I am prevented from moving properly or exercising, a lot of the other systems will degenerate – let alone my quality of life.
Musculoskeletal health is at the very center of longevity. In fact, to live a long, happy, active life, adequate health & function of the musculoskeletal apparatus are essential.
Tactics I follow to promote musculoskeletal health
- Keeping an eye on anabolic hormones
- Different forms of exercise
- Zone II cardio
- HIIT
- Resistance exercise
- Stability & mobility
- Interrupting sitting
- Experience with anabolics
- Healing injuries
Keeping an eye on anabolic hormones
A couple of years ago I was starving myself like Bryan Johnson, in part out of vanity, and in part because of online “health” advice applied in the wrong context. My IGF-1 was fairly low, my testosterone was very low, and my estradiol was abysmal. Doing a lot of (heavy) exercise in that state was a recipe for disaster, and I sustained repeated micro-injuries. Now, I make sure that my anabolic hormones (essentially IGF-1 & testosterone) are adequate to high-ish.
I am on my TRT lite regimen, which gives me testosterone and estradiol levels near the top of the reference range – discussed here. I also take a babydose of oxandrolone (discussed below).
Fortunately, since I vastly increased my caloric intake, my IGF-1 levels are naturally slightly above the reference range, at the top of the reference range for a 25-year old (which is probably the ideal range to keep for life). The upsides of IGF-1 include stronger immune function, greater vitality, less neurodegeneration, less “wear and tear” due to better repair systems, greater bone mass and muscle mass. The downsides include a slightly higher speed of aging (on paper) and a small increase in cancer risk. In my opinion, high IGF-1 levels blow low IGF-1 levels out of the water.
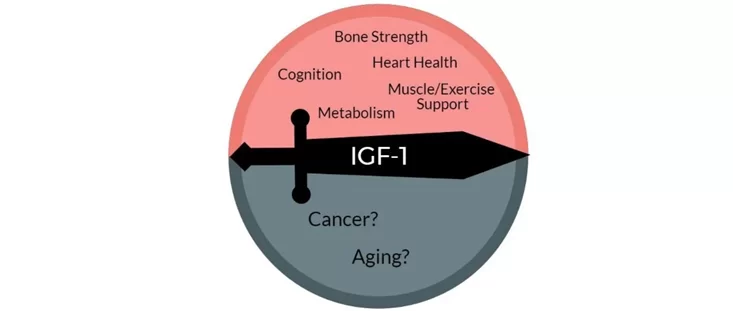
I engage in different forms of exercise
The body’s response to high-intensity interval training versus strength training versus purely aerobic exercise is very different. Therefore, I engage in different forms of exercise:
- resistance training (3 times per week)
- steady-state cardio in the zone II range either on an exercise bike or outdoor run (2x/w)
- brief high-intensity cardio after each zone II session (2x/w)
- occasionally sports games with friends (but I do not count these as exercise)
While some effects of the different kinds of exercise are universal and overlapping (e.g., improving metabolic health, brain health, cardiovascular health, sleep, and well-being), different kinds of exercise have non-overlapping individual effects as well.
For example:
- High-intensity exercise or HIIT induces antioxidant systems more than other forms of exercise.
- Low-intensity endurance exercise is much more than just “burning calories”. It specifically improves capillary density & flexibility, as well as mitochondrial number & function.
- Low-rep range heavy resistance training particularly improves myofibrillar density and contractility. It builds strength and mass.
- High-rep range resistance training leads to an increase in sarcoplasmic muscle growth and a host of muscle-related metabolic adaptations.
It is probably best to include all different forms of exercise, given the partially non-overlapping benefits. I discuss now all of them in a little more detail.
Zone II cardio
Zone II cardio improves mitochondrial health, metabolic health, and brain health. The idea is to train at low intensity (rule of thumb: heart rate of 180 minus age) for at least 40min per session.
Below is a screenshot from my Polar data of what a zone II session looks like for me (I usually end with a 5-minute high-intensity part). My average heart rate is between 70-80% of my maximum heart rate (which is a tad under 190bpm).

Zone II exercise is any type of cardiovascular exercise performed right at the cusp before lactate starts to accumulate. Said another way, it is exercise during which oxidation of fatty acids is at the maximum. Through a variety of signaling pathways, zone II exercise stimulates mitochondrial function and mitochondrial biogenesis, which vastly improves metabolic health through a variety of mechanisms.
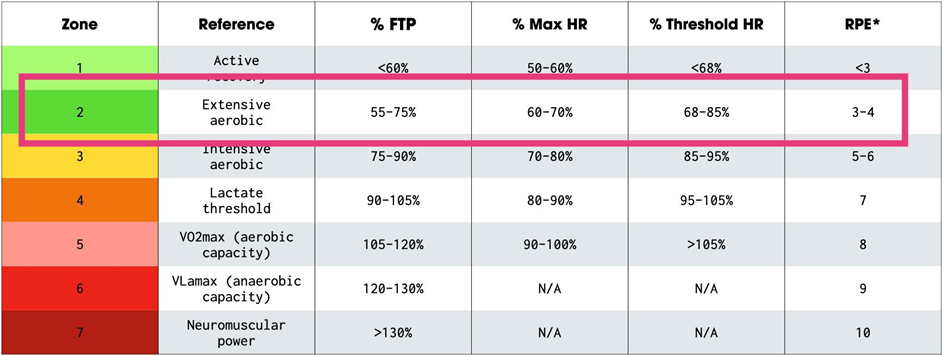
High-intensity exercise
I end each endurance session with a 5 minute high intensity session. It would probably be better to do a dedicated HIIT session once per week but I dread doing that. At the end of my 5 min high intensity session, I am sometimes near the point of absolute failure (“almost puking”). Repeating this 4x in a row? Fuck no. (The ideal high intensity session would look like: 4min all out effort –> 4 min low effort –> repeat 4x.)
So I find a single bout of a couple of high-intensity minutes a good tradeoff between “what works” and “what am I willing to stick to”. A medium program I stick to consistently is much better than a perfect program that I do not stick to.
It is thought that high-intensity exercise is great for optimizing neural health (e.g., BDNF & VEGF expression) and upregulating body-wide antioxidant systems due to the short oxidative burst during peak activity. Due to the principle of hormesis, this means that there will be less oxidative stress at baseline.
High-intensity sessions also improve VO2 max, which has a host of other benefits on pretty much everything related to health, longevity, and well-being.
Resistance exercise
Resistance exercise builds and maintains muscle mass. Besides the effect of muscle on how others perceive you (looks) and how you perceive yourself (self-esteem), muscle mass is highly correlated with strength, which is highly correlated with longevity and quality of life.
Moreover, the more muscle mass someone has when they are young, the more of it can be maintained into old age (maintaining muscle is easier than building it), which not only prevents falls and fractures but also increases quality of life and the range of activities someone is capable of doing.
Furthermore, next to looks and structural integrity, having a decent amount of muscle mass is also highly beneficial from a metabolic perspective, particularly in terms of insulin sensitivity.
I do some form of resistance exercise three to four times per week and I alternate between lower body and upper body days.
I usually do one to two drop-sets for each muscle group, totaling about 12-15 exercises per session. I start with the heaviest weight possible (for 4-6 reps) and go until (almost) failure. Then, without any rest, I immediately lower the weights by around 10% and go again until (almost) failure.
I repeat this process until I have decreased the weight by around 5-10 times. I believe that this gives me the best bang for the buck because it hits myofibrillar muscle growth + sarcoplasmic muscle growth + while also being a degenerate form of cardiovascular exercise.
Thanks to a low daily dose of metreleptin, which allows me to keep body fat levels low without suffering the neurocognitive and endocrine consequences of starvation, I am able to keep a muscular and lean physique pretty much year round. Below is “peak” condition but is not too far off from my “normal” state. In my normal state, I have about 2% bodyfat more but the same amount of muscle mass. This would not be possible with GLP-1 agonists as they are insufficient to fully counteract the hypoleptinemia associated with low body fat levels. I discuss this in more detail here: My Experience With Semaglutide.

A note on CrossFit
In the past I did CrossFit and I absolutely loved it. For me, CrossFit is a great “all-in-one”-solution as it combines stability, functional strength, low-intensity cardio, and a high-intensity metabolic conditioning part, all while being fun and social.

One major downside to CrossFit is that it gets a lot of people injured, and it might not be a great longevity sport. Fortunately, for the 1.5 years I have been doing it regularly I never got injured and interestingly, the knee pain I had before starting it has subsided. I only stopped because going to the CrossFit venue and back took too much time.
But I still sometimes miss CrossFit – especially the way it made me feel after. A good friend of mine says that before doing CrossFit his thoughts were 80% negative and 20% positive, and he claims that simply doing CrossFit flipped this ratio around.
Stability & mobility
At the end of my twenties, I can do a lot of dumb things incorrectly that will impair my joint integrity down the road.
Recently, I started to incorporate some more strategic forms of mobility training.
- I specifically strengthen my anterior tibialis (for knee health), my posterior chain (with a kettlebell), and my glute medius (with exercise bands), my hamstrings (in the form of nordic curls), and a couple of other “mobility” muscles. I specifically relied on the advice from Knee Over Toes Guy.
- Every day I stretch my hip flexors and the muscles of the quadriceps femoris, which tend to get tight.
Interrupting sitting
More of my day than I would like is usually spent sitting. I set a repeating alarm every two hours to incorporate multiple “exercise snacks”. These 2-minute-long bouts of exercise help me with alertness & cognition. My favorite exercise is kettlebell swings.
Kettlebell swings are great to spike my heart rate and to work my posterior chain, both of which are well-suited to counter some of the negative consequences of prolonged sitting.

Microdose of oxandrolone
Oxandrolone is an anabolic steroid. It is a DHT-derivative and therefore devoid of estrogenic and progesterone effects. Oxandrolone has about six times the anabolic potency of testosterone (anabolic-androgenic ratio of about 1:6 or so). Because it is not a substrate of 3α-hydroxysteroid dehydrogenase, it can exert its anabolic effects in skeletal muscle, unlike DHT, which is poorly active in skeletal muscle despite greater androgen receptor binding. DHT is broken down in muscle – as discussed here: My Experience With Finasteride
I take a microdose of oxandrolone (1.5mg/d) to help with adding some muscle (health benefits; vanity). While bodybuilders might find this dose laughable (as they generally use 10-50mg per day – often in combination with other anabolics), this was actually quite a decent anabolic boost considering that a healthy men´s natural T secretion is about 8-10mg per day (roughly 650-750ng/dl) and oxandrolone has an anabolic ratio of 1:6 – meaning that 1.25mg of oxandrolone (anabolic equivalent of about 6-8mg of testosterone) would almost double exposure to anabolic agents if my endogenous secretion did not decline (which is a big “if”).
I measured my blood three times. Even at this dose, oxandrolone slashed my LH, total testosterone, SHBG, and free testosterone by about 40% and, on paper, I was borderline hypogonadal (total testosterone of about 400ng/dl).
Interestingly, even such a microdose of oxandrolone caused me to shed about 1kg of water weight, which may have been due to a decrease in estradiol (or estradiol signaling). The combination of the slightly increased muscle mass and the slight loss of total body water caused me to look visibly more “cut” and bigger in a way that others were commenting on it, even though the scale was hardly affected.
While having no effects on liver enzymes and blood lipids, the effect on muscle mass was okay (about 2-3kg gain in lean tissue after about 3 months). However, I stopped due to its adverse effects on my endogenous sex hormones.
However, ever since hopping on my TRT lite regimen (which decouples my gonadal sex hormone secretion from hypothalamic input), I reintroduced it. On it, my liver enzymes and lipids are perfect, which would be my only concern. Last time I tested, my LDL-C was at 25mg/dl (I do take 5mg of ezetimibe).
Healing bones
If I ever happen to have a broken bone or ruptured tendon, I will turn to teriparatide (PTH-analogue), which activates osteoblasts and chondroblasts, therefore slashing recovery time. It helps bones grow as well as tendons heal. I got this “tip” from an osteologist, who mentioned that a couple of his colleagues had used teriparatide after breaking their bones while skiing and that they (reportedly) all healed up like Wolverine.
I suppose that the use of teriparatide is possibly widespread in professional sports Why else would professional athletes be so quick to be back on track? Hint: It is presumably not just massage therapy. Interestingly, neither the bro-science nor the bodybuilding community has gotten hold of this yet, while the use of potentially dangerous, questionably effective, and unregulated peptides with weird artificially scientific sounding names seems to be widespread. I discuss this in more detail here: Why I Do Not Use Peptides
My weak points
2 years ago (03/2024), I dislocated my left shoulder during a stupid iceskating stunt. I needed to go in for surgery and I recovered exceptionally well. However, unfortunately, I got reinjured twice (handstands before I was ready; aggressive chest flies), and ever since, my shoulder has not felt the same. I am currently taking a break from dedicated shoulder and chest training.
I also have easily inflammable patellar tendons, which I destroyed by running very fast with very bad shoes and terrible form (“heal striking”) for many years between the ages of 17 and 25. I am currently in the process of remodeling my tendon with the use of strategic isometrics.
Sources & further information:
- Podcast: Peter Attia – The importance of muscle mass, strength, and cardiorespiratory fitness for longevity
- Podcast: Peter Attia & Mike Joyner – Exercise, VO2 max, and longevity
- Scientific article: Muscle mass index as a predictor of longevity in older adults
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part IX: My Protocol for Optimizing Organ Health
I have discussed the difference between “cellular” longevity and organ health in Part II & Part III of my article on longevity.
If one of my organ systems gives in in isolation, even if all other things were otherwise impeccable, this would cause rapid and massive ripple effects eventually leading to body-wide impairment.
The usual causes of organ degeneration are the following:
- infarction (e.g., pulmonary degeneration after thromboembolism)
- infection (e.g., HBV targeting the liver)
- toxicity (e.g., tadalafil leading to ototoxicity)
- cancer (e.g., liver cancer)
Tactics I follow to support & monitor organ health
- General health
- Monitoring blood markers
- Regenerative medicine
General health
Obviously, many of the things discussed in My Longevity Protocol (Long & Technical Version) will help with maintaining my organs’s health and integrity. These include lowering the mTOR pathway, optimizing metabolic health, keeping my CVS as functional as possible, maintaining high-ish levels of anabolic hormones, lowering inflammation, preventing metals from accumulating, and trying to prevent cancer.
Monitoring blood markers
I monitor many organ markers and am ready to intervene accordingly including liver enzymes (ALT, AST, GGT), uric acid, creatinine, cystatin C, bone mineral density, spermiogram, hematological parameters, hormone markers, CRP, fibrinogen, tumor markers.
Regenerative medicine
Eventually, one or more of my organs will be impaired. Barring existential catastrophe or premature death, in a couple of decades I plan on using various approaches of regenerative medicine if they happen to be available and given we have enough data. These potentially include stem cell replacement, tissue engineering, nanotechnology, and gene therapies. I discuss some of these in more detail here: The Exciting Future of Longevity Medicine
Sources & further information
- Podcast: Andrew Huberman & Peter Attia – Exercise, Nutrition, Hormones for Vitality & Longevity
- Scientific article: Ranking Biomarkers of Aging by Citation Profiling and Effort Scoring
- Scientific article: The ageing kidney: Molecular mechanisms and clinical implications
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part X: My Protocol for Avoiding Toxins & Minimizing Infections
Part 1. Minimizing infections
A couple of years ago, my liver enzymes were elevated 10x above the upper limit of the normal reference range on a routine blood test. I did not have any symptoms whatsoever. Two weeks later, they were back to normal. The elevation of liver enzymes was presumably due to an asymptomatic viral infection.
My body is under continuous attack from microorganisms. Most of them won’t succeed in infecting me, but some of them will – often without me being aware of it.
The common cold is the most frequent infectious disease in humans. The average adult gets two to three colds a year. Over 200 viral strains can cause it, with rhinoviruses (and now perhaps SARS-CoV-2) being the most common.
Most of the centenarians say that they rarely got sick throughout their lives. In part because very healthy people rarely get sick but also in part because infections, and the ensuing immune response, wreak a lot of havoc throughout many bodily tissues accelerating the aging process.
Tactics I follow to prevent & treat infections
- Thyroid hormones
- Hygiene & sanitation
- Herpes viruses
- Cytomegaly virus
- Periodically killing sprees
- If I had to choose an antibiotic for infections
Thyroid hormones
A couple of years ago, I weaned off thyroid hormones to gauge how I feel without them. In the first month, I got a minor but noticeable infection three times, despite not having been sick for a couple of years prior. I restarted therapy and (noticeable) infections stopped. In part because of this, I increase my T3 dose by 6.25mcg in the winter.
I am currently off exogenous thyroid hormones.
Hygiene & sanitation during childhood
I grew up in a mountainous area. As a kid, I spent a lot of time outside, either playing in the woods or helping at my grandparents’ farm. It is possible that this lack of sterility had benefitted the development and “training” of my immune system.
Herpes viruses
Most humans are infected with at least two (usually more) herpes viruses. Herpes viruses are retroviruses, which means that they reverse-transcribe their genetic material, which is then integrated into human DNA. These viruses then remain latent in certain tissues throughout the body and constantly try to “break out”.

As I age and the human immune system gets weaker (immunosenescence), the ability of herpes viruses to break out gets better. A sizeable share of the human immune system is constantly trying to fight back these herpes viruses, usually without us noticing.
In fact, it is thought that in the elderly about 40-50% of the adaptive immune system is constantly trying to fight EBV and particularly CMV. Needless to say, infection with herpes viruses is associated with “inflammaging” as well as dementia.
For this reason, a good friend uses 500mg of valacyclovir daily (and plans on being on it indefinitely) aiming to keep viral load and constant reinfection of healthy cells low. Whether the upsides of taking valacyclovir outweigh the risks is currently unknown, though valacyclovir’s safety profile seems to be quite good.
Cytomegaly-virus (CMV)
In contrast to most other herpes viruses, CMV infects a variety of different tissues. It is not only associated with immunosenescence but also with body-wide systemic inflammation, ranging from osteoporosis to hearing loss to dementia. Fortunately, I am currently negative for CMV.
I use 500mg valacyclovir before a date (in case we get intimate) aiming to prevent infection with herpes viruses, particularly HSV-II and CMV. Unfortunately, valacyclovir is not very effective against CMV but it is the only herpes antiviral I am willing to take given that ganciclovir can increase the mutation rate in humans. Once a vaccine is available and proven to be safe, I am eager to get it.
Interestingly, there is evidence that rapamycin prevents CMV infection, possibly by reducing lymphocyte clonal expansion.
Periodically killing some of the organisms that may have set up shop in or on my body
Every other year, I do a short course of doxycycline (14d; 100mg) to clear my organism of some exotic, unwanted co-inhabitants (e.g., protists, exotic bacteria), followed by a short course of itraconazole (7d; 100mg) to do the same with fungal cells, followed by a short course of mebendazole (4d; 100mg) to do the same with all kinds of worms (particularly helminths).
If I had to choose an antibiotic for infections
Obviously, antibiotics are a huge spectrum and the “one-size-fits-all” antibiotics are broad-spectrum antibiotics (which is not good as they kill off most of the healthy flora). For most uncomplicated infections I think macrolide antibiotics are a great choice, particularly roxithromycin. Roxithromycin is dubbed “the doctor’s antibiotic” for a reason. It has a long half-life and needs to be taken only once daily. It targets intracellular bacteria as well (e.g., mycoplasma).
Furthermore, it also kills senescent fibroblast and is, after dasatinib, perhaps one of the most powerful clinically available senolytic drugs. Furthermore, unlike penicillin and cephalosporine antibiotics, it is relatively narrow-spectrum and does not wreak havoc on the gut flora.
Doxycycline is also a great choice, as is minocycline, which has the additional advantage of inhibiting microglia, therefore helping with neuroinflammation (and potentially Long-COVID). Both are relatively easy on the gut microbiome. Furthermore, both antibiotics also kill off potential unrecognized co-inhabitants that for most people go unnoticed (such as symptom-free infections with intracellular parasites).
For fungal infections (e.g., yeast), first-line therapy is usually fluconazole. I prefer itraconazole over fluconazole because itraconazole does not penetrate well into the CNS because it is a substrate of p-glycoprotein. I prefer not to expose my brain to these kinds of drugs unless necessary.
Sources & further information
- Website: Wikipedia – Hygiene Hypothesis
- Scientific article: Hygiene hypothesis
- Scientific article: Cytomegalovirus infection is associated with increased mortality in the older population
Part 2. Avoiding toxins
Whether I like it or not, there is an ongoing accumulation of toxic waste products in all tissues of my body. These include metals, microplastics, and the accumulation of undegradable byproducts (e.g., lipofuscin).
Tactics aimed at avoiding toxin exposure (hint: none!)
In an ideal world, there would be sunscreens only made of non-absorbable pigments, ways to prevent the accumulation of microplastics, consumer products devoid of endocrine disruptors, and water filters that remove lead and other toxic metals. Unfortunately, I do not live in an ideal world. The next best place currently is the EU, which does a decent job at regulating consumer health (at least in comparison to other countries).
Currently, there are only few strategies I follow:
- not drinking warm/hot drinks out of plastic bottles
- not using cosmetic products (other than lip balm and sunscreen)
- testing my blood levels of lead every other year or so
- not eating salmon (which accumulates a lot of shitty stuff due to biomagnification)
- avoiding overly processed food whenever possible
Other than through the strategies above, I pay little attention to (potential) toxins encountered in everyday life because that would limit my freedom too much for uncertain value. I may change this in the future as more data arrives and/or easier ways to limit toxin exposure become available.
Sources & further information:
- Scientific article: Loss of life expectancy from air pollution compared to other risk factors: a worldwide perspective
- Scientific article: A Detailed Review Study on Potential Effects of Microplastics and Additives of Concern on Human Health
- Scientific article: Food additives in childhood: a review on consumption and health consequences
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part XI: Optimizing Mental Health
Mental health is the strongest predictor of life satisfaction and explains twice as much as the second strongest factor (physical health).

Tactics I follow to promote mental health
- Vitality
- Sleep
- Human connection
- Purpose
- Mindset
- Sex
- Gratitude
- Meditation
- Exercise
- Supplementation
- Neuropharmaceutical
Vitality
I believe that the most important condition underlying mental health is “baseline vitality” (energy levels, mood, health). Improving physical health will improve mental health as the mind emerges from the brain, which is part of the body. I discuss baseline vitality here.
Sleep
- Good sleep improves all aspects of my health & well-being. If my sleep is crap, everything is crap.
- For me, regularity is key. Going to bed at the same time every day (+/- 30 minutes) improves all aspects of my sleep.
- Light is picked up by the retina and transmitted to the brainstem, where it interferes with sleep even if I do not consciously notice it. I use blackout curtains & magnetic tape.
- The same is true for sounds. Noises impair deep sleep even if I am not aware of it. Using earplugs has considerably improved my sleep quality.
- Whenever I go to bed in a cold environment, I fall asleep faster, and I sleep better. Year-round I use a cooling blanket made of nylon and polyethylene. I also use sheets made of silk, which keeps my body cooler.
- I expose myself to bright blue light in the morning. I have a custom-built setup that produces 60.000 lux at a distance of 3m (about 6x as much as light therapy lamps at a distance of 30cm). Will post about in the future. This not only helps with the entrainment of my circadian rhythm, but also improves my sleep, hormones, energy levels, and mood.
- In the past, I abused melatonin as a sleeping pill. However, I stopped using it as it might blunt the secretion of multiple other hormones.
- Before sleep, I take 5 grams of glycine and 400mg of magnesium, both of which improve my deep sleep slightly.
- Occasionally, I use a low dose of a sleeping pill. At the moment, I use daridorexant perhaps once a month and a microdose of zolpidem whenever I have a night-shift in the hospital.
Human connection
I am looking to be loved, to be appreciated, to matter. And a lot of things I think and do are probably rooted in this core desire, which is deeply ingrained in human biology and survival instincts.
Throughout our evolutionary history, a member of Homo sapiens left by itself was doomed to die since it did not have access to tribe-provided stuff such as food, shelter, warmth, protection, and mates. Everything related to its survival was found within and through the social domain, which is still true today.

Therefore, evolutionarily speaking, belonging to a tight-knit group and having deep connections with others was (and still is) of existential importance to humans. When a human being lacks it, the brain evolved to respond with feelings of distress (loneliness) in the same way it evolved to respond with feelings of hunger and thirst whenever there is a shortage of food or water.
Conversely, humans who are part of a community are rewarded with feelings of deep well-being in an analogous way evolution rewarded humans with short-term pleasure from sugar or sex.
In a similar way that hypothalamic cells measure energy intake and determine appetite vs. satiety (POMC neurons), new research suggests that there is something similar for regulating “social satiety”, including “rebound socialization” after prolonged social isolation in rats.
This has been proven in rodent species and a certain group of hypothalamic neurons responsible for regulating social behaviors (MPV neurons) has been identified. These neurons are intimately connected to oxytocin and vasopressin neurons.
In the human brain in particular, large parts of the brain are specifically designed for dealing with social stimuli, hierarchy negotiation, status concerns, and keeping score of social relations.
Unfortunately, today’s society doesn’t facilitate the same kind of camaraderie and sense of community that humans are likely wired to require. The recent rise of individualism with a focus on status and self-esteem surely did not help. This has left many people feeling empty and lonely.

From the available research it seems that having high-quality social ties is associated with better health and a longer life expectancy – though admittedly the effect is probably not as direct as it may seem due to possible confounders being at play such as healthy and vital individuals being more likely to have high-quality connections.
Regardless of putative effects on health and lifespan, having high-quality relationships is an important factor for mental health and well-being, probably even exceeding exercise or sleep. Said another way, individuals with great human connections but “bad” sleep and exercise habits may be happier than individuals with poor human connections but otherwise “great” lifestyle habits.
As Mark Manson expresses it: “And just like a beaver must build a dam to truly express his beaverness, humans must form social bonds to fully express our humanity.” Or as Johann Hari puts it, just like bees evolved to live in a hive, we humans evolved to live in a tribe. If we disband our tribes, we pay a price.
In my early twenties, I used to be quite introverted and prone to spending most of my time on “my things”. Over the last couple of years, I was fortunate enough to find my tribe of people who I can connect with in a deep and genuine way.
Having found my community is one of my biggest sources of fulfillment and has improved my life, productivity, and happiness in ways that are difficult to overstate. In a way, I would call it my most important “biohack”.
A note on status
Related to connection & belonging is the intrinsic human desire for status. This desire is highly biologically determined because, for millions of years, someone’s status in society determined preferential access to food, mates, and shelter. In fact, large areas of the human brain are dedicated to status & hierarchy negotiation, both of which are of essential importance for survival and reproductive success.
So, it is no surprise that a change in status would lead to a change in certain aspects of brain function. In fact, your (perceived) status in society has a strong influence on your neurotransmitters and neuroendocrine function (hormones). For example, in many primate societies (including humans), after males rise in ranks and become alpha males, testosterone and cortisol levels are known to rise. Similarly, depressed individuals often perceive themselves to be low status (and vice versa) – something known as the “rank order hypothesis of depression”.
A modern manifestation of this inherent drive for status is people’s desire to be “popular” on social media and the self-worth alterations by other people liking (or not liking) your content.
In this way, status (or the perception of one’s status) affects energy levels and mood through its effects on neurotransmission and neuroendocrine function.
And being low status is usually connected with poor mental health – and there likely is also some causality involved.
“Rank-theory hypothesis”
Some researchers speculate that depression may have proved useful in dominance conflicts that are unlikely to be won. According to the “rank theory of depression”, sustained melancholy, withdrawal behavior, and a fixation on personal shortcomings and insufficiencies may ensure that the weak “keep their heads down” and don’t overreach themselves, which may prove evolutionarily fatal.
The rise of depression and anxiety in young people coincides with the advent of social media, and there is probably a causal connection at play. Social media constantly floods people with “evidence” that many others have it better. I like what Naval Ravikant has to say about it: “Social media make celebrities of all of us. And celebrities are the most miserable people on Earth.”
According to this theory, depression may represent a fitness-enhancing adaptation to group living. Indeed, if individuals are “losing” in life, they are more likely to develop depression. In support of this may also be the fact that adolescents are often depressed (because most adolescents are not on top of the hierarchy – aka the cool kids). Further support for this theory is evidence that antidepressants may overturn dominance hierarchies in relationships.

Mindset: The “Top-down” Component
I like to divide factors that influence my mind into “bottom-up” and “top-down”.
- “Bottom-up” factors are any biochemical changes that influence my mind, including neuropharmaceuticals, sleep, exercise, and hormones. Most of my blog is about bottom-up effects.
- “Top-down” factors are primary mental changes that then influence biochemistry but may also have non-biochemical effects.

In the following paragraphs, I describe my experience with top-down factors and the effects they had on my physiology.
On some days I am tired. If I then do something that excites me, such as having a deep conversation with a friend, it is always striking how this increases my energy and mood. In the same way that a good conversation can have a stimulant-like effect on me for a few hours, so can working on a project I burn for stimulate me for weeks to months. Biochemically, this is presumably mediated in part by a top-down mediated increase in a variety of neurotransmitters.
A few more examples:
- A decade ago, I used to be semi-depressed for about half a year or so because I was reducing myself, my thoughts, and my feelings to molecular biology. Then, simply learning about the concept of emergence was quite powerful in terms of boosting my liveliness. In other words, I boosted my liveliness from “within” the mind.
- The few times I fell in love with someone, I was always amphetamine-like stimulated for a couple of weeks, sometimes months. I was euphoric, needed less sleep, was less hungry, and had more energy. The neurobiochemical cascade of falling in love is characterized by higher levels of oxytocin, glutamate, and dopamine, among other things. Interestingly, falling in love is known to increase cortisol secretion by 40-100%.
- Before meeting one of my ex-girlfriends, I was in quite a bad place mentally. I was lonely, restless, and my “Urvertrauen” (trust in the Universe that things will be alright) was gone. I then saw somebody I was blown away by. I approached her and asked her out. On our first date, I knew that we would be a couple soon. After this date, my Urvertrauen was restored and all the restlessness, loneliness, and unease – that had been there fore months – was gone immediately, as if somebody had flipped a switch in my brain – even though my hormones or monoamines had not changed much. This change in how I felt was holding up for months. This was quite eye-opening to my bottom-up centered worldview.
- A couple of years ago, after one of my ex-girlfriends broke up with me, I was devastated for many months. In the same way that falling in love with her caused a hypomania-like state, losing her led to a state resembling true biochemical depression. The neurobiology of grief is thought to be characterized by an altered monoamine transmission and a dysregulated neuroendocrine control (hormones), among other things.
- In the past, days before a major exam, I was often super stressed, and many aspects of my mental and biochemical functioning were altered. These included heart rate changes, increased sweating, and even altered gut motility.
A few more non-personal examples:
- The placebo effect is a prime example of a top-down effect. The placebo group in antidepressant studies usually measurably improves in a variety of domains. Similarly, being hopeful or simply believing in something can have measurable physio-biochemical ramifications.
- For many people, unresolved trauma can have massive effects on neurobiology, endocrine function, and physiology – sometimes for years.
- Children that are mistreated or neglected sometimes fail to thrive (to grow properly). This form of “psychosocial dwarfism” can occur even when adequate caloric intake is provided.
- In orangutan tribes (orangutans are somewhat closely related to humans), there is only one alpha male and something about the sheer presence of an alpha male has other male orangutans growth-suppressed (it is thought that his screams play a role). After the alpha male dies, the growth-suppressed young orangutans suddenly go through puberty. This means that puberty was induced from “within the mind”. And the alpha male grows two flangs on the side of his cheeks. Something in his brain is sensing his alpha-male status leading to specific physiological changes that are reserved for alpha males.
In all of these instances, biochemical changes are induced “top-down” (from within the mind) solely due to “information”. It is therefore not too far-fetched to claim that, for example, having a pessimistic or nihilistic mindset can sustainably alter monoamine transmission and hormones, and presumably things that are even “deeper” (e.g., gene expression), negatively (which then in turn favors a nihilistic or angst-ridden mindset).
On the other hand, it has been shown that positive thinking, gratitude, optimism, or “purpose” can have striking effects on neurobiology, endocrine function, and physiology. (This is not to say that biochemical enhancement is not useful in finding purpose, love, or developing an optimistic mindset in the first place.)
Andrew Huberman and Jordan Peterson discuss the influence of “purpose” and “meaning” on dopaminergic neurotransmission here.
The main point of this entry is basically that mental health can be quite deeply affected by not just changes that are bottom-up (e.g., altering hormones or neurotransmitters trough lifestyle of exogenous molecules) but also by more “psychological” changes (i.e., information), which eventually affect deep material processes within the brain, and presumably also within individual cells. While this may seem “duh” to most people, for me, a biological materialist at heart, this was quite eye-opening to find out about – even though the biological mechanisms are far from being elucidated.
Purpose
After experimenting with all kinds of molecules, I found that, like human connection, purpose is the best antidepressant (reducing suffering & making me happy) as well as stimulant (giving me loads of energy & motivation).

Throughout most of my life, I had a sense of purpose, and whenever I was lacking it, vitality enhancement, with a special focus on dopamine always seemed to increase my chances of finding it again. However, out of all of the things on this website, purpose is probably the hardest one to get.
Here, I want to specifically mention psychedelics, which helped some of my friends a great deal in this regard. Next to having helped with getting unstuck in the egocentricity loop (“I am the center of the universe”), a friend who was gripped by a sense of meaninglessness came out of one session feeling part of something larger than himself. The effect catalyzed an upward spiral.
However, I also know people who were seriously harmed by psychedelics so they are best approached with the respect they deserve.
I discuss psychedelics in much more detail here.
Gratitude
For the past couple of months or so, I started doing a very simple 2-minute gratitude exercise whenever I enter a public transport vehicle, which is roughly twice per day.
I close my eyes and start to think of 5 things I am grateful for:
- #1: Something beautiful that happened today (e.g., That I had a great conversation while going for a walk with a friend.)
- #2: Something beautiful that is about to come later today (e.g., Going for my evening gym session.)
- #3: Something concerning my personal relationships (e.g., That I get along well with my siblings and that I even have them.)
- #4: Something big in my life (e.g., That I am allowed to work in a field that I am intensely interested in.)
- #5: Something beautiful this very moment (e.g., being without physical pain – something that is not as obvious as it seems for non-medical professionals)
The human brain is a pattern recognition machinery, trained to find patterns for things it is frequently used for. For example, if I train myself to look for people with red hair, it won’t be long before I see red-haired people everywhere. If I frequently play chess, my brain gets better at finding chess patterns.
Similarly, if I frequently think about things I could be grateful for, my brain gets better at “gratitude” simply because the responsible neural networks are trained every day to find “patterns” of things I could be grateful for.
The most important thing is that I really feel these things so that the emotions leave neural traces/imprints. I highly recommend the book hardwiring happiness.
Over time, these thoughts of gratitude came on much more readily – either spontaneously at random times throughout the day or specifically as an antidote to stress, worrying, or grudging. In both cases, these thoughts of gratitude reliably lift my mood and increase my well-being. This small habit of gratitude may be the single best habit I do in terms of effort and payoff.

Meditation
I meditate daily for 5-10 minutes, which is probably way too little. Meditation (supposedly) not only increases my emotional health, but it presumably also has a number of measurable longevity benefits.
- Firstly, the gain in the ability to concentrate decreases rumination by the simple fact that my mind jumps less. Since “a wandering mind is an unhappy mind”, this may have measurable benefits for cortisol levels, sleep, and blood pressure.
- Secondly, meditation increases mindfulness. The increase in mindfulness allows me to be less on autopilot and to increase my “window of agency” by not being constantly pulled in a million directions. This has implications for habits and behavior in general, both of which are tightly linked to the lifestyle choices I make and do not make on a daily basis.
Sex
Having good sex (for me, most important is emotional intimacy) is an important pillar of my well-being. A loss of sexual ability (e.g., Peronyie disease, severe sexual dysfunction, disfiguring injury) would therefore be a huge loss. I will post about penis health in the future.
Exercise
Exercise is great for brain health and well-being. Different forms of exercise have partially overlapping and partially independent effects. Benefits include a feeling of accomplishment, mitochondrial health, neurogenesis, upregulation of serotonergic & opioidergic tone for hours after, the social aspects, and positive feedback of looks on self-worth.
As with sleep, the time invested in exercise has a high ROI on a number of domains, including my mental health, energy, mood, creativity, looks, and happiness. Furthermore, exercise is among the healthiest things I can do for my brain. In this section, I will discuss the effects of exercise on vitality and mental health only.
There is a neurobiochemical reason for the saying “You are only one workout away from a good mood.”.
- Firstly, during intense exercise, the “nasty” kappa-opioid system in the nucleus accumbens shell (the brain’s major hedonic hotspot) is activated, which mediates the subjective experience of discomfort. However, as a counterregulatory mechanism, the “feel-good” mu-opioid system is activated and remains activated for a couple of hours after
- Secondly, exercise boosts noradrenaline and cortisol, both of which increase central nervous system “readiness” for a couple of hours.
- Thirdly, exercise increases serotonin transmission for many hours to come. Whenever vertebrates exert themselves intensely, serotonin levels rise, in part to signal to the animal “Hey, chill down.” This prolonged rise in serotonin, which presumably serves self-protective purposes, is another main reason intense exercise makes me feel good for the entire day.
- Fourthly, exercising makes people proud of themselves and gives people a feeling of accomplishment, which is also important for mental health.
These neurotransmitters are discussed in more detail here: An Introduction to Neurotransmitters
Exercise makes me feel and function so well that I would even do it if it did not have any other beneficial long-term effects – which it undoubtedly has.

I particularly appreciate its beneficial effects on both short-term brain function as well as long-term brain health. My nervous system is the basis of everything I think, do, and feel, and few things improve it as much as regular exercise.
- Exercise stimulates the release of nerve growth factors such as BDNF & VEGF. In fact, it seems to be one of the most neurogenetic interventions available to mankind – endurance exercise more so than resistance training.
- Regular vigorous exercise promotes cell proliferation in the hippocampus, which enhances mood, memory, and cognition. In part because of this, exercise is probably among the most powerful tools I have to stave off cognitive decline.
- Exercise stimulates the locus coeruleus, the brain’s major source of noradrenaline, and causes this collection of cells to increase in size and baseline activity. Therefore, over time, exercise really does lead to a more alert brain and the reason is a long-term structural change (or better, a combination of multiple structural changes).
- Exercise increases brain oxygenation and improves sleep, both of which are crucial to brain health.
Through the combination of these short-term and long-term effects on the brain, exercising regularly improves energy levels, mood, and cognition, and also slows the rate of neurocognitive decline.
There are all sorts of other reasons (beyond brain health) why exercise is powerful beyond belief. Exercise improves sleep, hormones, insulin regulation, nutrient partitioning, and musculoskeletal health. Exercise is also one of the most potent interventions available to slow neurocognitive decline.
Regular vigorous exercise also causes hundreds of changes in gene expression, affecting almost any level of physiology, biochemistry, metabolism, and health.
If family or friends ask me about the single most important thing they can do to improve their health and vitality, my answer is always the same: sleep & exercise. Of note, the exercise recommendations by official health agencies are pathetically insufficient for it to have the vast benefits it could have.
Supplementation
I make sure I do not have any vitamin or mineral deficiency. Furthermore, I take a couple of supplements that might have favorable effects on mental health. I discuss the supplements I take, and why I take them, in more detail here.
Neuropharmaceuticals
A few years ago, I started using the adaptogens ashwagandha and rhodiola during a time I was very stressed. Because I had already decided to go down this route (namely trying to alter my mood with the introduction of foreign molecules), from an ethical perspective the argument was already settled.
To me, there is no difference between natural (“good”) vs. synthetic (“evil”) molecules, so I decided to switch to neuropharmaceuticals instead. Given that the modern world is a minefield and not very conducive to mental health, in my opinion, living in the modern world is just as unnatural as using pharmaceuticals to better navigate it.
I experimented with numerous neuropharmaceuticals, but few to none stuck. I describe the neuropharmaceuticals I experimented with in more detail here.
Sources & further information
- Opinion article: What Kind of Antidepressant Should I Take?
- Scientific article: Can meditation slow rate of cellular aging? Cognitive stress, mindfulness, and telomeres
- Scientific article: The efficacy and safety of nutrient supplements in the treatment of mental disorders: a meta-review of meta-analyses of randomized controlled trials
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part XII: What Genetic Tests Told Me About My Health & Longevity
Most centenarians today are centenarians because of their exceptional genes.
A couple of years ago I decided to get my genetics tested. I bought kits from 23andMe, My Heritage, and Ancestry. I briefly looked at their reports but found them worthless. However, all three companies provide access to the raw data, which I then merged into a single file. I uploaded this to various online services, the most important of which was Promethease.
Obviously, genetic tests are to be taken with a huge grain of salt because the association between allele and phenotype often has a lot of confounding variables. Nonetheless, I found out a couple of interesting things, some of which had actionable insights.
Longevity
I have good versions of SIRT1 (hype created by David Sinclair), FOXO3 (common in centenarians), and CDK2ND (cyclin-dependent kinases are important in cell division), all of which are uncommon in the Caucasian general population, and all of which are associated with longevity.
Self-decode puts my genome-wide association longevity score at the 99th percentile, so I was ultra-lucky in this department (given that their percentiles have actual predictive power). Last year, my grandpa died at the age of 96 with no hint of dementia, cancer, or heart attack.
According to another genome-wide association study (GWAS), I am at the 97th percentile for genes associated with long-standing health without frailty.
Dementia
I have no APOE4 allele but two APOE3 alleles. A single copy of that gene increases the risk of Alzheimer’s by 300%. Two copies by roughly 1500%. Individuals with two copies get AD almost 90% of the time. Conversely, individuals with two E2 alleles get AD less than 1% of the time. Furthermore, I am homozygous for a “good” version of CETP (cholesterol-ester transfer protein) that is associated with a markedly reduced risk for dementia (20% of the population).
This means that my genetic risk for Alzheimer’s disease (the most common form of dementia) is somewhat low. I discuss my dementia prevention protocol here.
Cardiovascular disease
No adverse polymorphism in any lipoprotein-associated genes (e.g., Lp(a), LDL-R, PCSK9 function, HMG-Co-Reductase), which means that my risk for heart disease is not artificially elevated. Genetically, I am at the 15th percentile of Lp(a) levels. Lp(a) is the particle of death, and is highly associated with heart attack, stroke, and amputations.
Unfortunately, I have a Factor V Leiden mutation, which means that my risk for thromboembolism is higher than the general population. I discuss my atherosclerosis prevention protocol here.
Cancer
I have no deleterious polymorphisms in JAK2 (tyrosine kinase important in cell division), AKT1 (upstream of mTOR), and a beneficial polymorphism in p53 (cell cycle break; associated with a three-year longer lifespan; elephants have dozens of copies and thus get less cancer despite greater cell mass), which means that my genetic risk of cancer is on the lower end. However, my mum had breast cancer, and my dad had prostate cancer, so I do not know how much of this is true. Cancer prevention protocol here.
Diet
I have “normal” PPAR alpha and PPAR gamma alleles, which are important for fatty acid metabolism and insulin sensitivity. Polymorphisms with these genes are associated with a poor tolerance of high-fat diets from a metabolic standpoint.
Autoimmune disease
I do not have the dreaded HLAB27 polymorphism (allele important for presenting “foreign” peptide fragments to the immune system), an allele often associated with a large number of of autoimmune diseases.
Inflammation
I am homozygous for a gene associated with low CRP, another gene associated with low IL-6 levels, and a gene associated with low IL-23 levels. I am not sure of its real-world significance. Article on how I combat inflammation here.
“Bad genes”
I was quite lucky in that department. No APOE4, bad CETP, bad genes associated with high Lp(a) or bad lipid markers, or bad genes associated with cancer or autoimmunity. No monogenic diseases. The only really bad gene I have is a factor V Leiden mutation, meaning that my risk of clotting is considerably higher than the rest of the population – about 5% of Europeans have that gene. However, on blood tests, my clotting times are usually slower than the end of the reference range, which is weird.
Other interesting or actionable insights
- I have a couple of “adverse” polymorphisms in the SLC2A9 gene coding for the urate transporter, which is associated with high uric acid levels. This was no surprise, as my uric acid levels were always on the higher end, and for which I had been taking 150mg of allopurinol already.
- I have a couple of polymorphisms associated with low vitamin D levels, and therefore increased my intake of vitamin D from 2000IU/d to 4000IU/d.
- I have two versions of the “sprinter gene” (ACTN3), which is uncommon in Caucasians (25%) but very common in people of African origin. It is associated with an increased number of fast-twitch muscle fibers and with a lower risk of muscle injury.
- I have a potentially deleterious allele in a NAC-transferase, which is associated with a 7x elevated risk for hearing loss. Therefore, I started to supplement with 300mg N-acetyl cysteine per day. My supplement protocol here.
- I am homozygous for three genes associated with a reduced ability to detect bitter, which means that I like vegetables more than people without these SNPs (all else being equal), which in turn is associated with a lower risk of obesity.
- I have a “bad” version of MTRR and two bad SNPS in MTHFR, both of which are associated with a “bad” methylation phenotype. Therefore, I supplement with 1mg L-methyl folate, and 1g TMG.
- I have a gene associated with poor conversion of ALA. Therefore, I increased my omega-3 fatty acid intake.
- I have a gene associated with a low synthesis of coenzyme Q10, which I now supplement with (60mg/d).
- I have a couple of genes associated with less hairiness. I have little body hair, so that checks out. Not even a year of TRT increased my body hair by any significant degree.
- I am homozygous for an allele (ABCC11) associated with less body odor and dry ear wax, which means that my risk for infections of my outer ear canal is low when I wear ear plugs.
- I have two interesting SNPs associated with interleukins. Firstly, I have a SNP that is strongly associated with low IL-6 levels. I am also homozygous for an SNP (1%) associated with low IL-23 levels, which are associated with lower CRP levels.
- I am homozygous for a version of CETP (cholesterol-ester transfer protein) that is associated with a markedly reduced risk for dementia (20% of the population). CETP inhibitors are currently being developed for atherosclerosis.
- I have a normal MAO-A gene (so no “warrior gene”), but with moclobemide, I can mimic the phenotype.
- I have normal JAK2 versions (leukemia/lymphoma risk). Janus kinase inhibitors are drugs that are used for autoimmune diseases or leukemia/lymphoma.
- I have multiple genes associated with an increased risk of prostate cancer. My use of finasteride is thus not just good for reducing my rate of cosmetic aging but also for reducing my risk of prostate cancer.
I discuss the influence of genetics in more detail here: Biohacking Can’t Beat Genetics
Sources & further information
- Scientific article: Review and meta-analysis of genetic polymorphisms associated with exceptional human longevity
- Scientific article: Analysis of Polymorphisms in 59 Potential Candidate Genes for Association With Human Longevity
- Podcast: Peter Attia – AMA #8: DNA tests, longevity genes, metformin, fasting markers, salt, inflammation, and more
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Part XIII: Side Effects of my Longevity Protocol
In this article, I´ll briefly discuss some roadblocks and side effects I have encountered from experimenting with healthspan interventions.
Liver and kidneys
Is all of the stuff I do and take bad for my liver and kidneys? My levels of cystatin C have improved and ALT/AST are always well within the normal range (both below 25 U/l). The only thing that consistently spiked my liver enzymes was acarbose.
Drug side effects
- Metformin: Initially, I could not function due to fatigue, which went away after one to two months of use. However, the bloating and indigestion did not.
- SGLT-2 inhibitors: I had to pee a lot. Furthermore, I was constantly dehydrated, which was the reason I stopped taking them. I discuss my experience with metformin and SGLT-2 inhibitors in more detail here.
- Semaglutide: I had some slight morning nausea for the first couple of weeks. Furthermore, I lost some of the “pleasure” associated with eating (which though, for me, is not an unwanted effect). I discuss my experience with semaglutide in more detail here.
- Senolytics: Flu-like symptoms & diarrhea on the first and second day. Dexibuprofen helped somewhat. I discuss my experience with senolytics in more detail here.
- Rapamycin: Initially some canker sores, and pitting edema in my legs. Also, for the first few months, my fasting blood sugar was elevated (100-120mg/dl). I discuss my experience with rapamycin in more detail here.
Knee pain
I did have one potentially major adverse effect, which could have been due to high-dose rapamycin during my first year on it (10mg/w). After 6 months or so, I started to get some knee pain in both of my knees. However, I had started doing regular sprints at the same time I had started rapamycin, so it was impossible to say whether the rapamycin caused (or at least exacerbated) it.
Nonetheless, the knee pain could have been related to rapamycin’s inhibitory effect on tissue repair and anabolism (its negative effects on wound healing are well-known). While the knee pain did not get worse for many months, it also did not go away completely until I had stopped taking the rapamycin for three months. An MRI scan revealed no signs of pathology.
It could have been a form of patellofemoral syndrome (runners’ knee), or it could have been something else. I do not know, and probably never will. Because of this, I decreased my rapamycin dose from 10mg to 5mg and now I also incorporate a 1-2 month break every year.
If the knee pain was indeed caused by rapamycin and if it did not have gone away (which it fortunately did), then this single side effect (i.e., developing degenerative knee changes for a long time to come) would have canceled out all of the potential benefits of rapamycin (and perhaps even all of the rest) because a potential inability to properly exercise would do a lot of damage to my body overall.
I am flying blind
Every time I have talked to doctors and longevity scientists, I realized that a lot of what longevity medicine does is far from established by “hard” science. Even though there is a vast amount of theoretical knowledge on the subject, when it comes to evaluating objectively how my interventions are performing, there are few to no useful ways to test for it.

Even though I track a long list of blood markers, body composition via DEXA, metabolic health via CGM, and do a full-body MRI every couple of years, I have little to no clue whether my interventions are actually doing something, or even worse, whether they are doing more harm than good.
What complicates the problem is the impossibility of getting truly objective markers. For example, I recently tested for FGF23. Because the marker was included in two different panels, by mistake, I tested for it twice within a 10-minute period. The delta between the two blood draws was quite large (39pg/ml vs. 50pg/ml on a reference range of 32-95pg/ml), despite both samples having been taken literally simultaneously and despite both having used the same assay.
Unfortunately, most of the objective data available is not only corrupted by biological and laboratory fluctuations but assays from one company to the next are also not comparable to one another. And neither are individuals.
All this leaves me with the following problem:
- When it comes to evaluating the effects on biological age, I have little idea what to truly test for. Every method available to date (e.g., telomere length, Howarth clock, glycan score) has severe limitations. For example, a multi-day fast is known to set back the Howarth clock by multiple years. Or excessive exercise is known to increase glycan age by increasing levels of tissue inflammation.
- Blood markers have a lot of problems. Individuals vastly differ from one another, in part due to genetic and endocrine differences, which then influence laboratory parameters in ways unrelated to underlying health status. Furthermore, essays between different companies differ. These issues make it hard to impossible to compare myself to other individuals, and at times also to compare myself to myself.
In sum, I am flying blind.
Part XIV: Cost, Effort, Risks, Benefits, “Why”
Cost
About 800$ per month (including food). As I was still on metreleptin, the cost was much more.
Effort
It might seem that adhering to my regimen takes a lot of effort. It does not.
- I prepare pillboxes about once per month (1h), and take my supps & drugs two times per day (morning; before bed) with a glass of water.
- Injecting HCG & metreleptin (roughly 1 minute per day)
- Exercise (including my way there and back) (1.5 hours per day)
- Preparing foods (20-30min per day) – I am all for convenience (e.g., Kefir+ olive oil)
- Extensive blood work every 3 months
Developing and fine-tuning everything took me years to do, but once it had been set up, it now takes me an average 5 minutes per day (excluding exercise and preparing food).

How did this start?
Due to being too much into “fitness”, I developed stubborn hormone problems at the age of 21, followed by medically supervised hormone replacement therapy. After experiencing the extent to which exogenous intervention was able to transform my outer and inner life, I started to go down the rabbit hole of biological self-optimization.
Slowly, one at a time, and with a deep understanding of how things work, I experimented with a lot of different things. I kept about 5% of the things I experimented with.
Am I worried about long-term consequences?
For some of my interventions, I do not have the proper long-term data I would like to have (i.e., in the context of a healthy person). So yes, I am worried about the long-term consequences of my actions. However, I am equally if not more worried about the long-term consequences of inaction.
(Potential) Benefits
All of the biomarkers I test for are great, though most of them have also been great before. I look young for my age, I feel well, I am athletic, I have good energy levels, and I can work in a focused way for long hours. But so are many others my age. Thus far, I have not seen any obvious benefits of my regimen. However, encouragingly, I have also not experienced any obvious downsides over the course of over half a decade.
My protocol is probably far from sufficient to meaningfully counter the aging process. Rather than extending maximum lifespan, it may have a much stronger effect on my health span, hopefully allowing me to live in a state of reasonable vitality until my time in this universe has run out.
Goals
My objective is to strike a decent balance between longevity and vitality (as unfortunately, longevity and vitality are sometimes at odds – for example, caloric restriction). I personally do not care about maximum life span extension as this comes with quality of life tradeoffs I am not willing to make. Said in other words, my objective is to live well for a long time (health span).
If I had to choose between longevity and vitality, I would choose vitality without a second thought. Fortunately, these two are not necessarily mutually exclusive. Longevity is not only about prolonging health span, but also about being healthy and feeling good in the here and now because improving metabolic, cellular, and tissue function presumably also benefits me in how I feel and function in my everyday life.
Risks
Many people perceive what I do to be aggressive, dangerous, detrimental, risky, and stupid. However, I believe that most of the things I do are much less aggressive, dangerous, detrimental, risky, or stupid compared to e.g., alcoholism, tobacco abuse, or obesity, which affect about 50% of the population.
In my opinion, the major difference between these things and what I do is the so-called “familiarity principle”: The more we are exposed to something (e.g. alcohol, cigarettes, obesity), the more familiar we are with it – and the more familiar we are with something, the more normal and safe it seems. However, my cells could not care less about arbitrary culture-bound familiarity.
Even though some of this is uncharted territory (at least it was when I first started doing it five years ago), I believe that the risks of my current regimen are quite low given the currently available clinical and experimental data. The riskiest thing I did was most likely CrossFit.
Furthermore, I continuously monitor a number of things, I do a full-body MRI every three years, and I have ready access to a lot of great doctors. People who are doing anything similar without extensive self- and other-monitoring, particularly if said people do not have a medical background, are likely heading for catastrophe.
Side effects
I have been on this or a similar protocol for about five years. No major side effects as of yet (see above).
Philosophy
I briefly describe my philosophy, and why I do everything I do, here.
Longevity Protocols
- My General Longevity Framework
- My Most Likely Cause of Death – My Protocol for Fighting Atherosclerosis
- The Ultimate Frontier – My Protocol for Fighting Cancer
- My Protocol for Fighting Dementia – What I Do To Keep Brain Health Optimal
- My Approach to Hormone Optimization for Longevity
- Crucially Important & Underrated – My Protocol for Optimizing Metabolic Health
- What Is All the Fuss About mTOR?! – Why (and How) I Reduce the Activity of the mTOR Pathway
- Eliminating Zombie Cells – My Protocol for Fighting Senescent Cells
- Burning to Death – My Protocol for Lowering Inflammation
- My Physical Fitness Protocol
- My Protocol For Optimizing Organ Health
- Avoiding Toxins & Minimizing Infections
- My Protocol For Optimizing Mental Health
- What Genetic Tests Told Me About My Longevity
- Side Effects of My Longevity Protocol
Subscribe to the Desmolysium newsletter and get access to three exclusive articles!
Disclaimer
The content on this website represents the opinion and personal experience of the author and does not constitute medical advice. The author does not endorse the use of supplements, pharmaceutical drugs, or hormones without a doctor’s supervision. The content presented is exclusively for informational and entertainment purposes. Never disregard professional medical advice or delay in seeking it because of something you have read on the internet.
